ES
Executive Summary
The ocean is essential for all aspects of human well-being and livelihood. It provides key services like climate regulation, through the energy budget, carbon cycle and nutrient cycle. The ocean is the home of biodiversity ranging from microbes to marine mammals that form a wide variety of ecosystems in open pelagic and coastal ocean.
Observations: Climate-related trends, impacts, adaptation
Carbon emissions from human activities are causing ocean warming, acidification and oxygen loss with some evidence of changes in nutrient cycling and primary production. The warming ocean is affecting marine organisms at multiple trophic levels, impacting fisheries with implications for food production and human communities. Concerns regarding the effectiveness of existing ocean and fisheries governance have already been reported, highlighting the need for timely mitigation and adaptation responses.
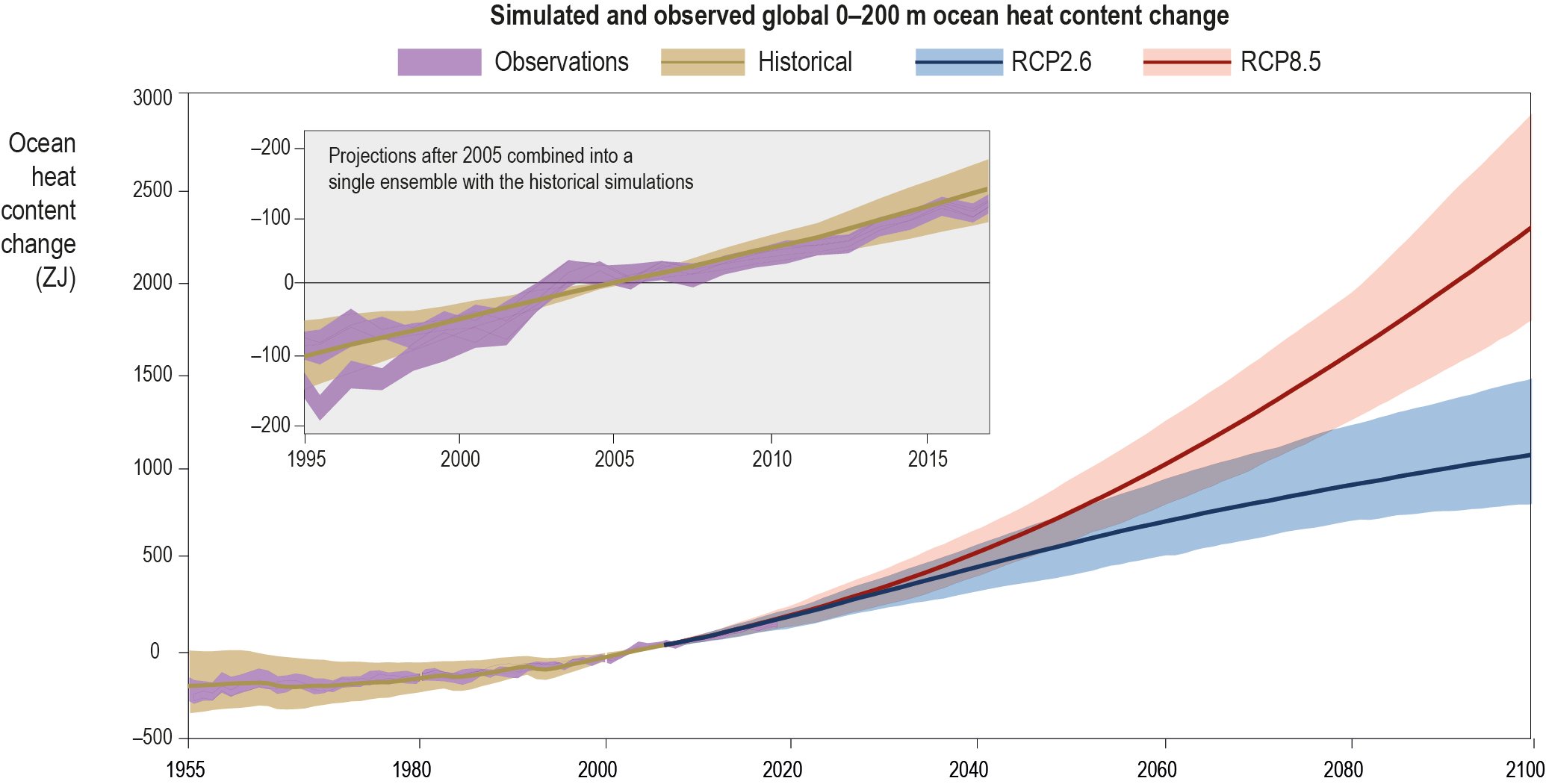
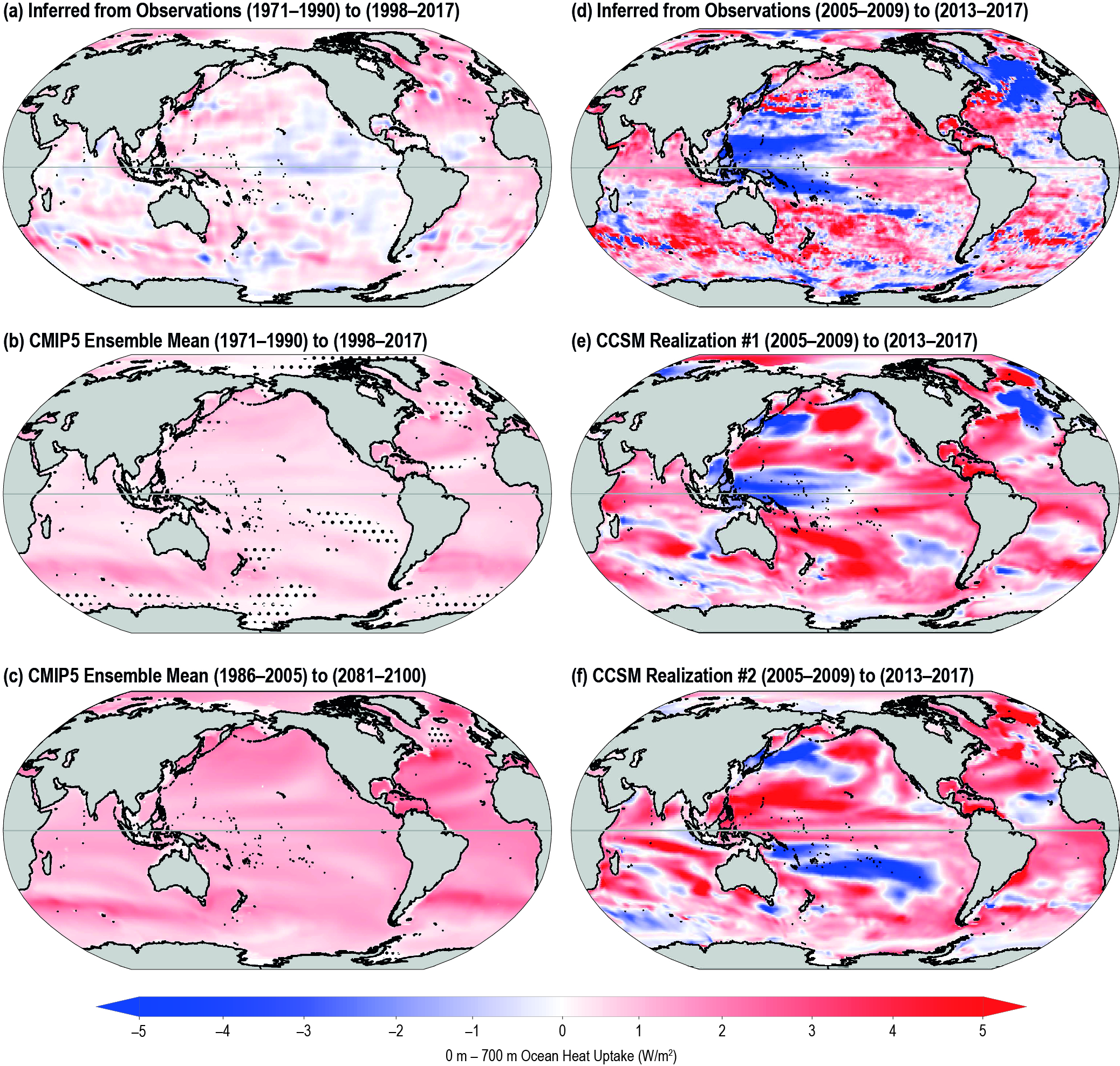
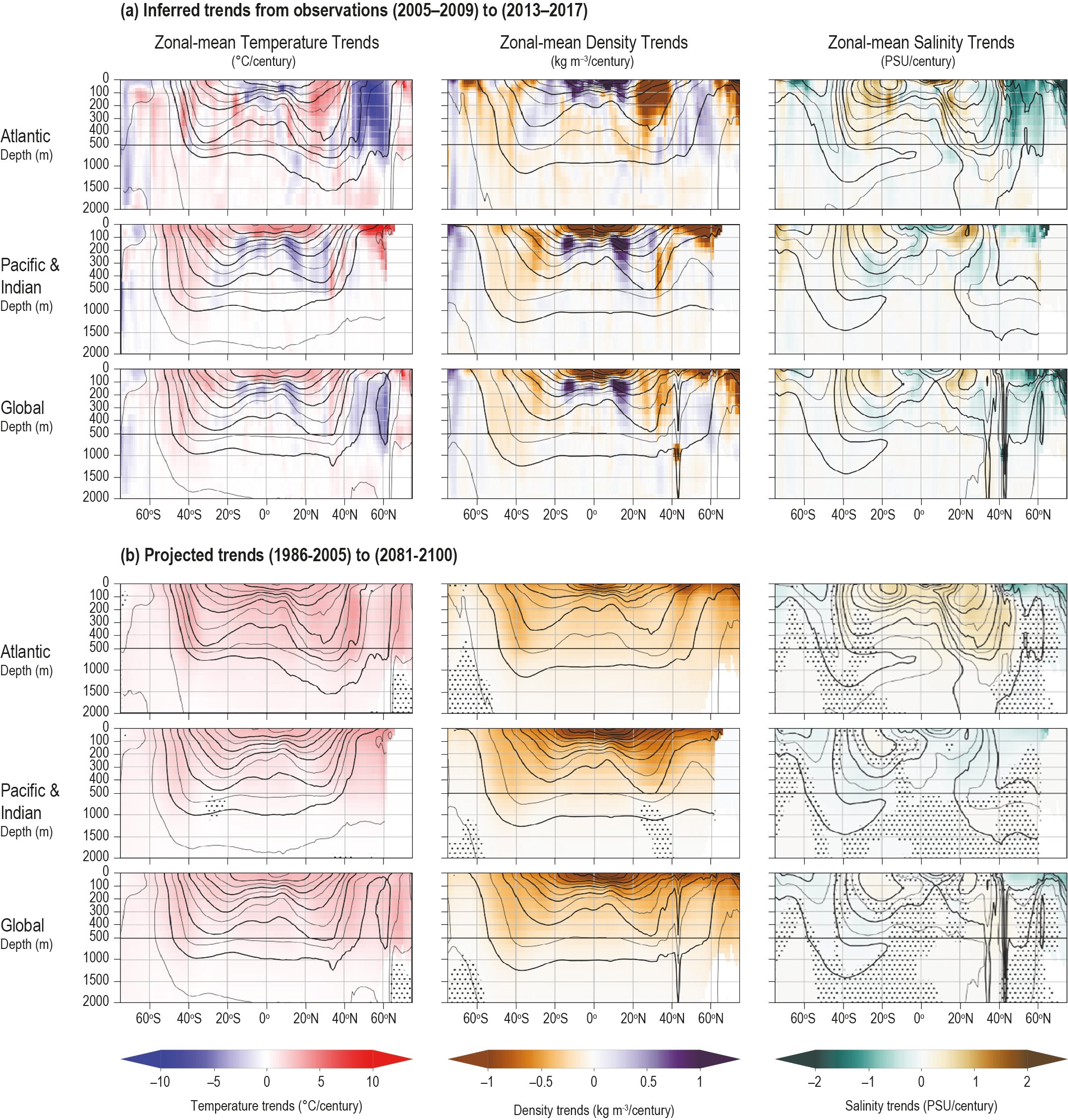
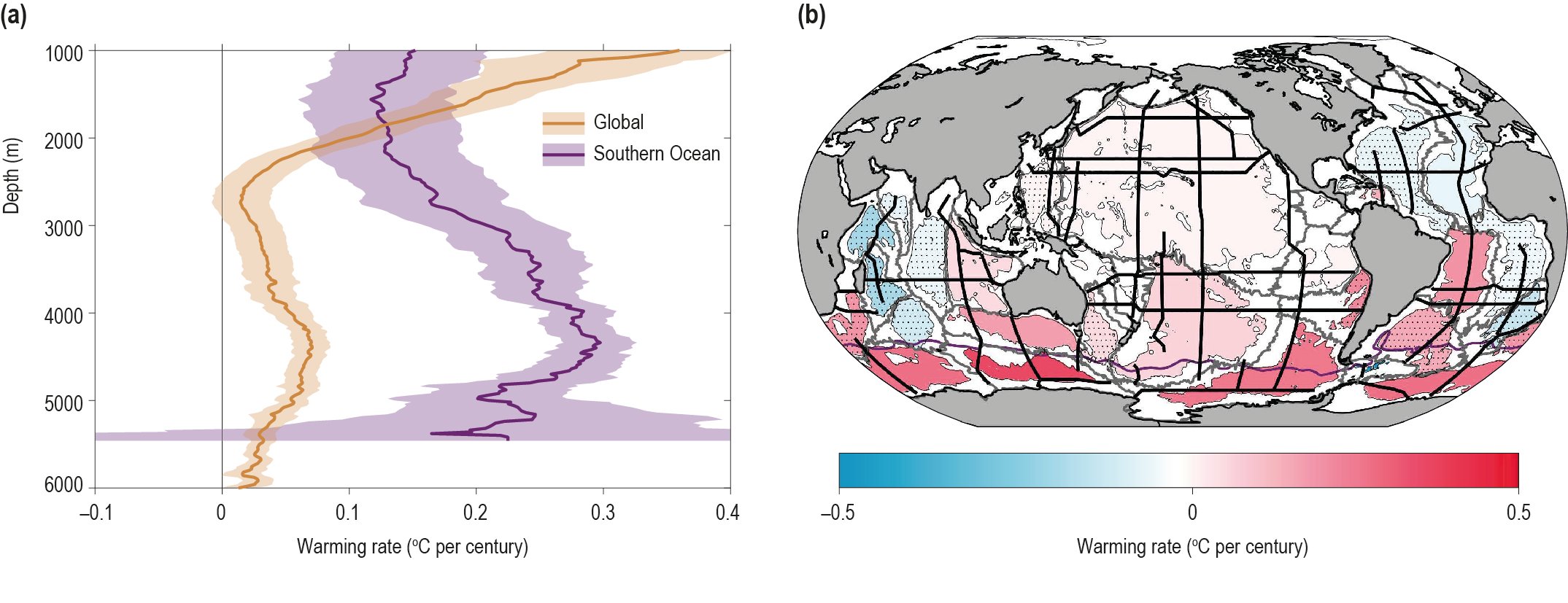
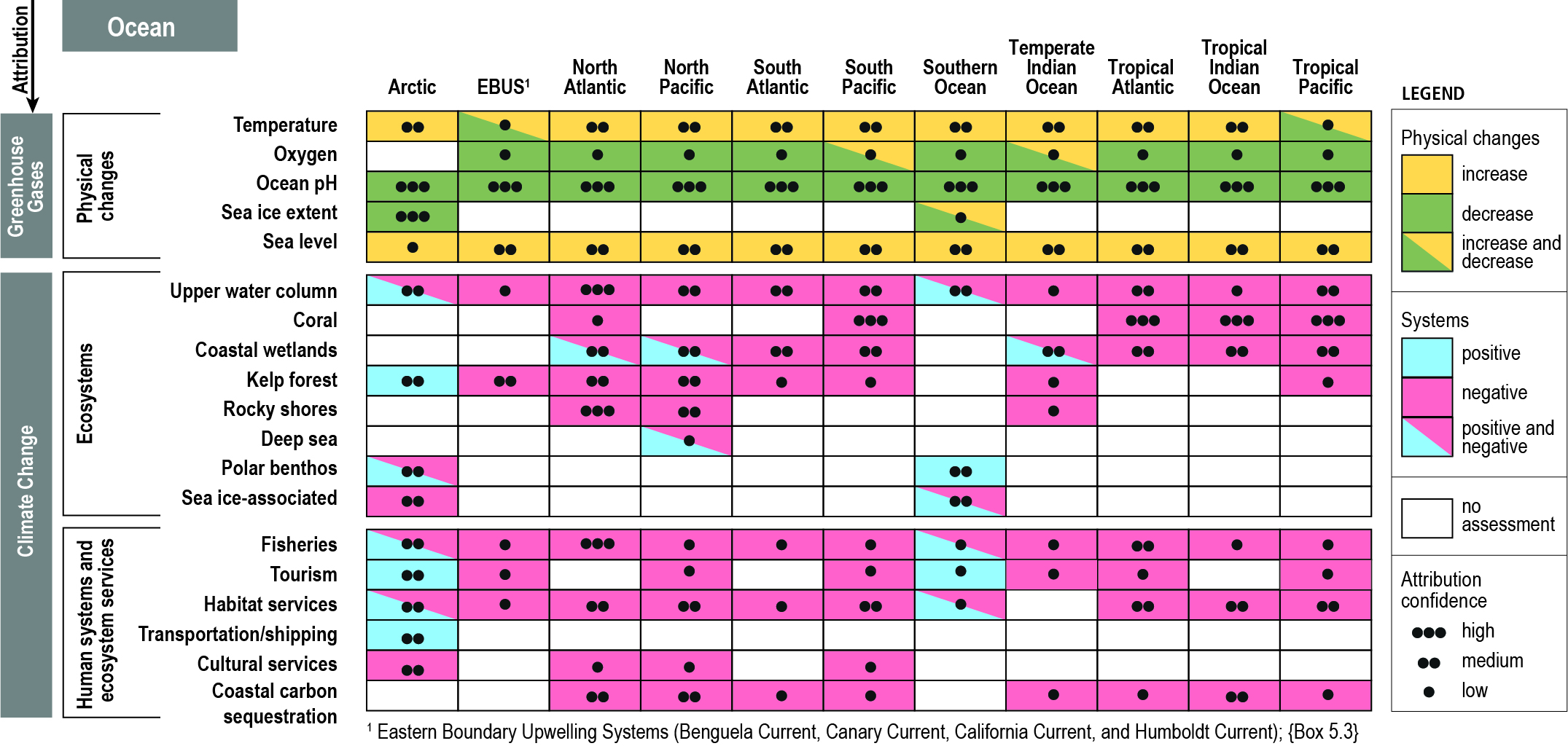
The ocean has warmed unabated since 2005, continuing the clear multi-decadal ocean warming trends documented in the IPCC Fifth Assessment Report (AR5). The warming trend is further confirmed by the improved ocean temperature measurements over the last decade. The 0−700 m and 700–2000 m layers of the ocean have warmed at rates of 5.31 ± 0.48 and 4.02 ± 0.97 ZJ yr-1 from 2005 to 2017. The long-term trend for 0–700 m and 700–2000 m layers have warmed 4.35 ± 0.8 and 2.25 ± 0.64 ZJ yr-1 from between the averages of 1971-1990 and 1998-2017 and is attributed to anthropogenic influences. It is likely 1 the ocean warming has continued in the abyssal and deep ocean below 2000 m (southern hemisphere and Southern Ocean). {1.8.1, 1.2, 5.2.2}
It is likely that the rate of ocean warming has increased since 1993. The 0−700 m and 700–2000 m layers of the ocean have warmed by 3.22 ± 1.61 ZJ and 0.97 ± 0.64 ZJ from 1969to 1993, and 6.28 ± 0.48 ZJ and 3.86 ± 2.09 ZJ from 1993 to 2017. This represents at least a two-fold increase in heat uptake. {Table 5.1, 5.2.2}
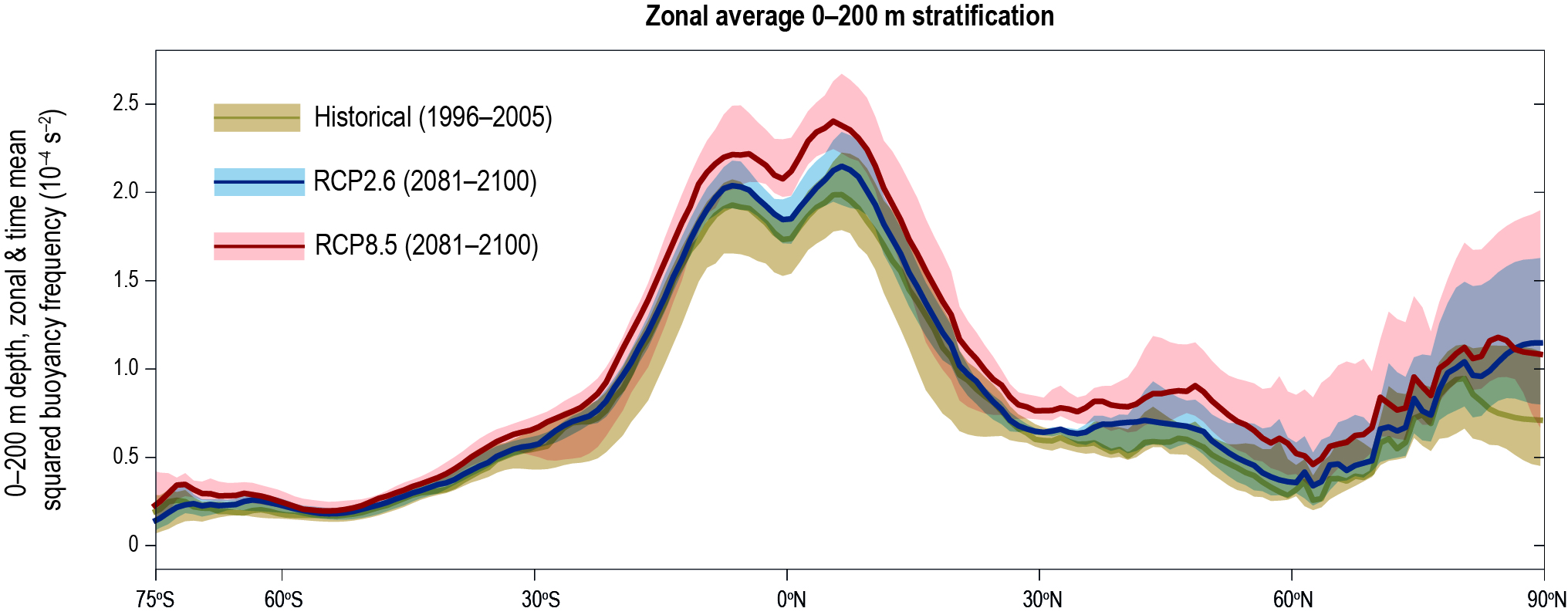
The upper ocean is very likely to have been stratifying since 1970. Observed warming and high-latitude freshening are making the surface ocean less dense over time relative to the deeper ocean (high confidence 2) and inhibiting the exchange between surface and deep waters. The upper 200 m stratification increase is in the very likely range of between 2.18−2.42% from 1970 to 2017. {5.2.2}
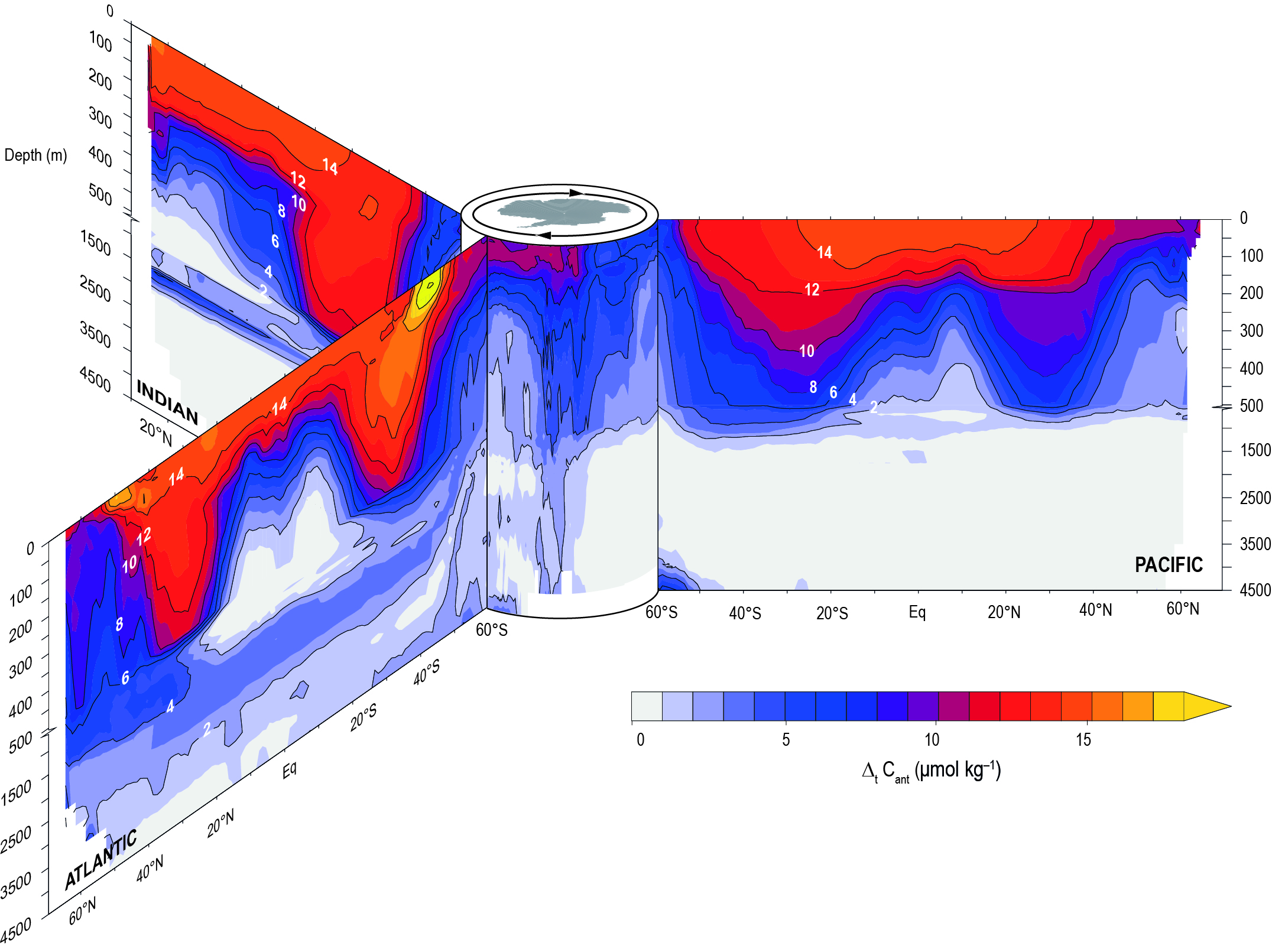
Multiple datasets and models show that the rate of ocean uptake of atmospheric CO2 has continued to strengthen in the recent two decades in response to the increasing concentration of CO2 in the atmosphere. The very likely range for ocean uptake is between 20–30% of total anthropogenic emissions in the recent two decades. Evidence is growing that the ocean carbon sink is dynamic on decadal timescales, especially in the Southern Ocean, which has affected the total global ocean carbon sink (medium confidence). {5.2.2.3}
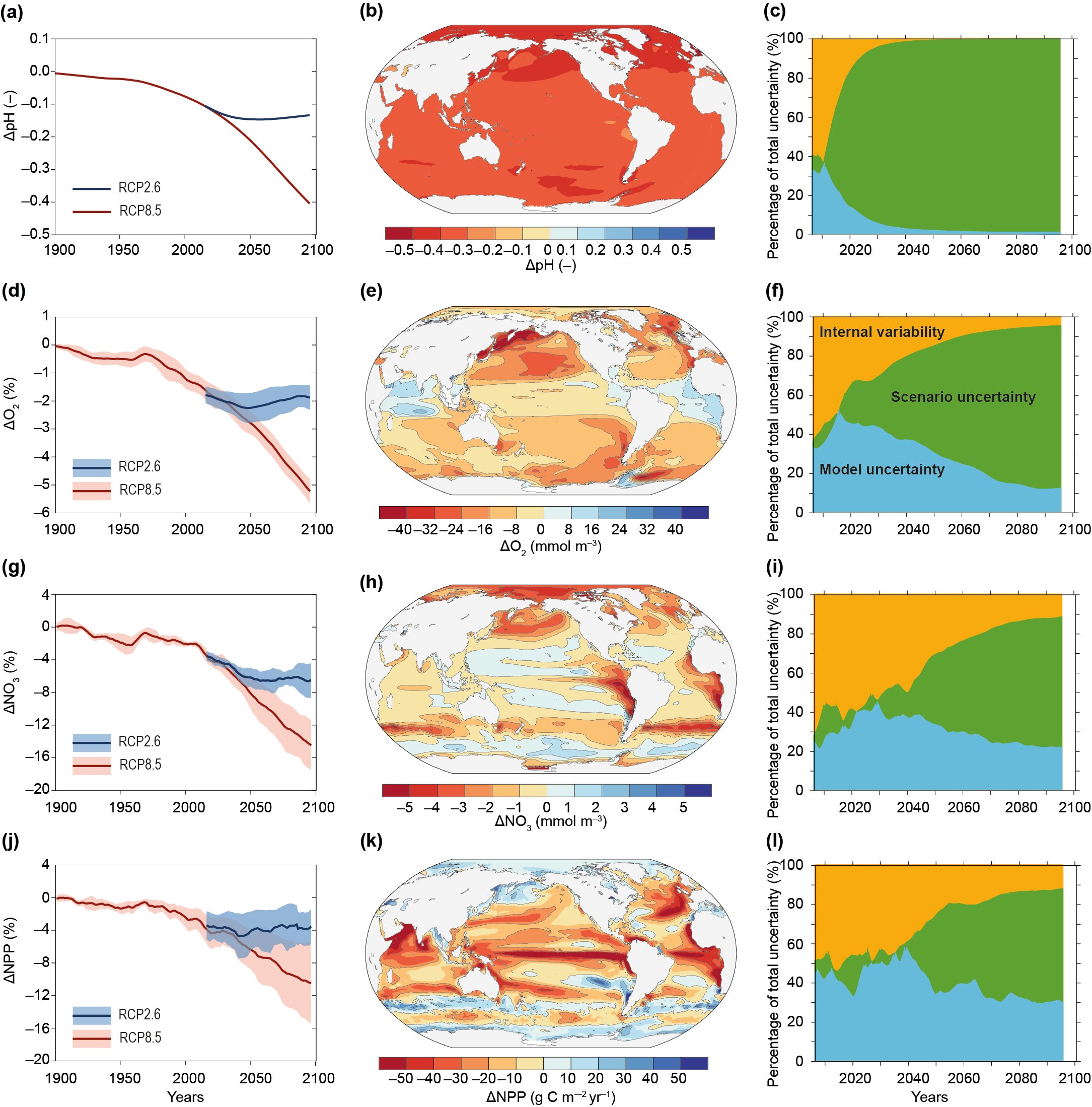
The ocean is continuing to acidify in response to ongoing ocean carbon uptake. The open ocean surface water pH is observed to be declining (virtually certain) by a very likely range of 0.017−0.027 pH units per decade since the late 1980s across individual time series observations longer than 15 years. The anthropogenic pH signal is very likely to have emerged for three-quarters of the near-surface open ocean prior to 1950 and it is very likely that over 95% of the near surface open ocean has already been affected. These changes in pH have reduced the stability of mineral forms of calcium carbonate due to a lowering of carbonate ion concentrations, most notably in the upwelling and high-latitude regions of the ocean. {5.2.2.3, Box 5.1}
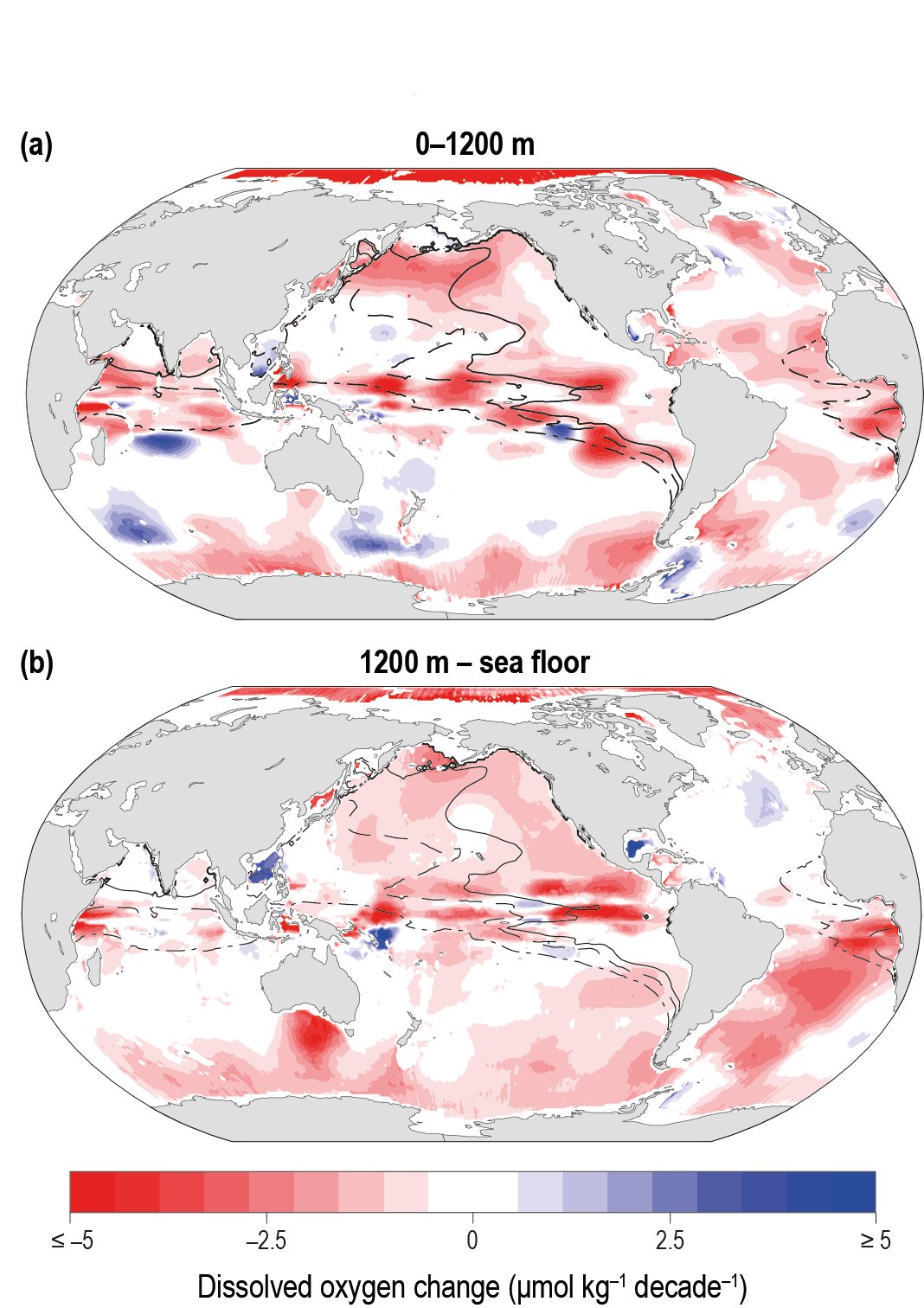
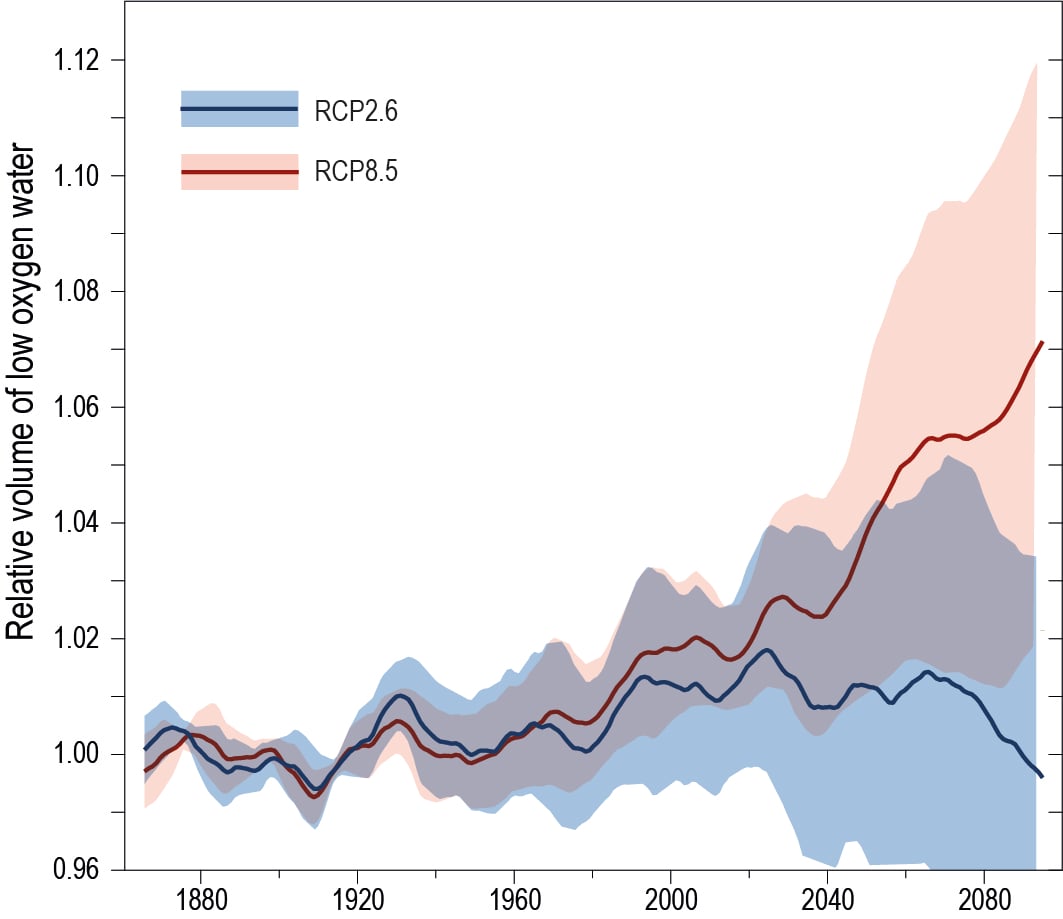
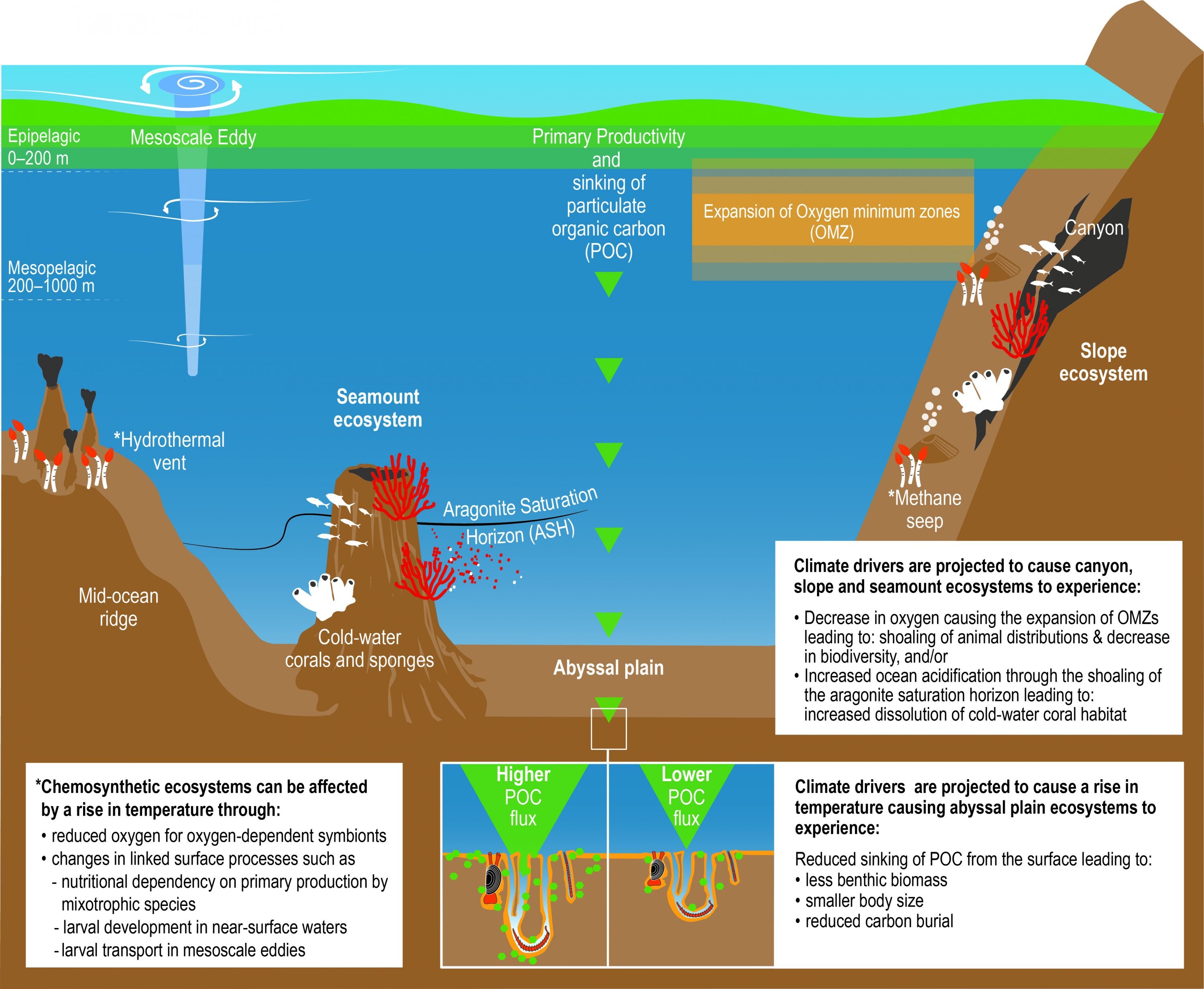
There is a growing consensus that the open ocean is losing oxygen overall with a very likely loss of 0.5−3.3% between 1970–2010 from the ocean surface to 1000 m (medium confidence). Globally, the oxygen loss due to warming is reinforced by other processes associated with ocean physics and biogeochemistry, which cause the majority of the observed oxygen decline (high confidence). The oxygen minimum zones (OMZs) are expanding by a very likely range of 3–8%, most notably in the tropical oceans, but there is substantial decadal variability that affects the attribution of the overall oxygen declines to human activity in tropical regions (high confidence). {5.2.2.4}
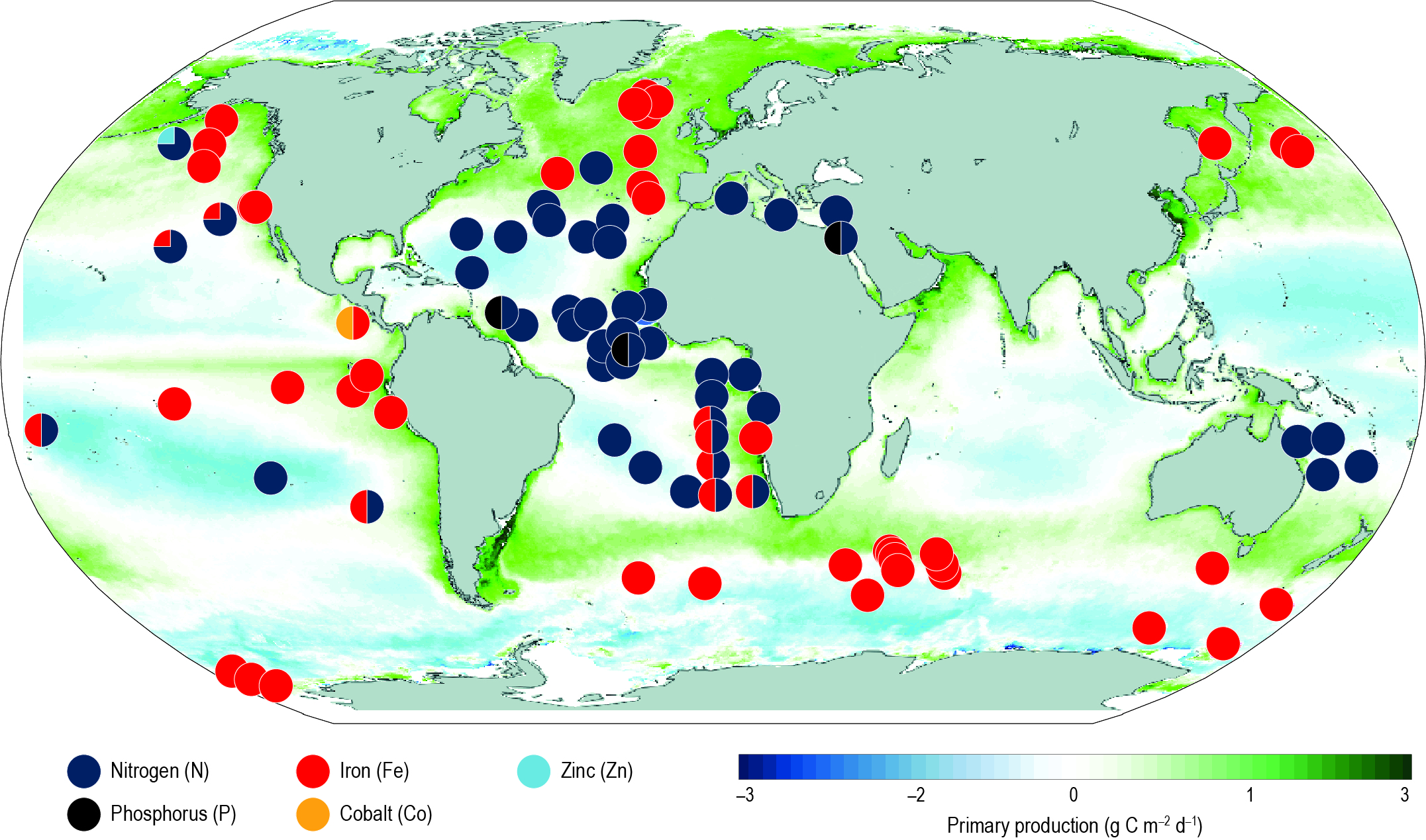
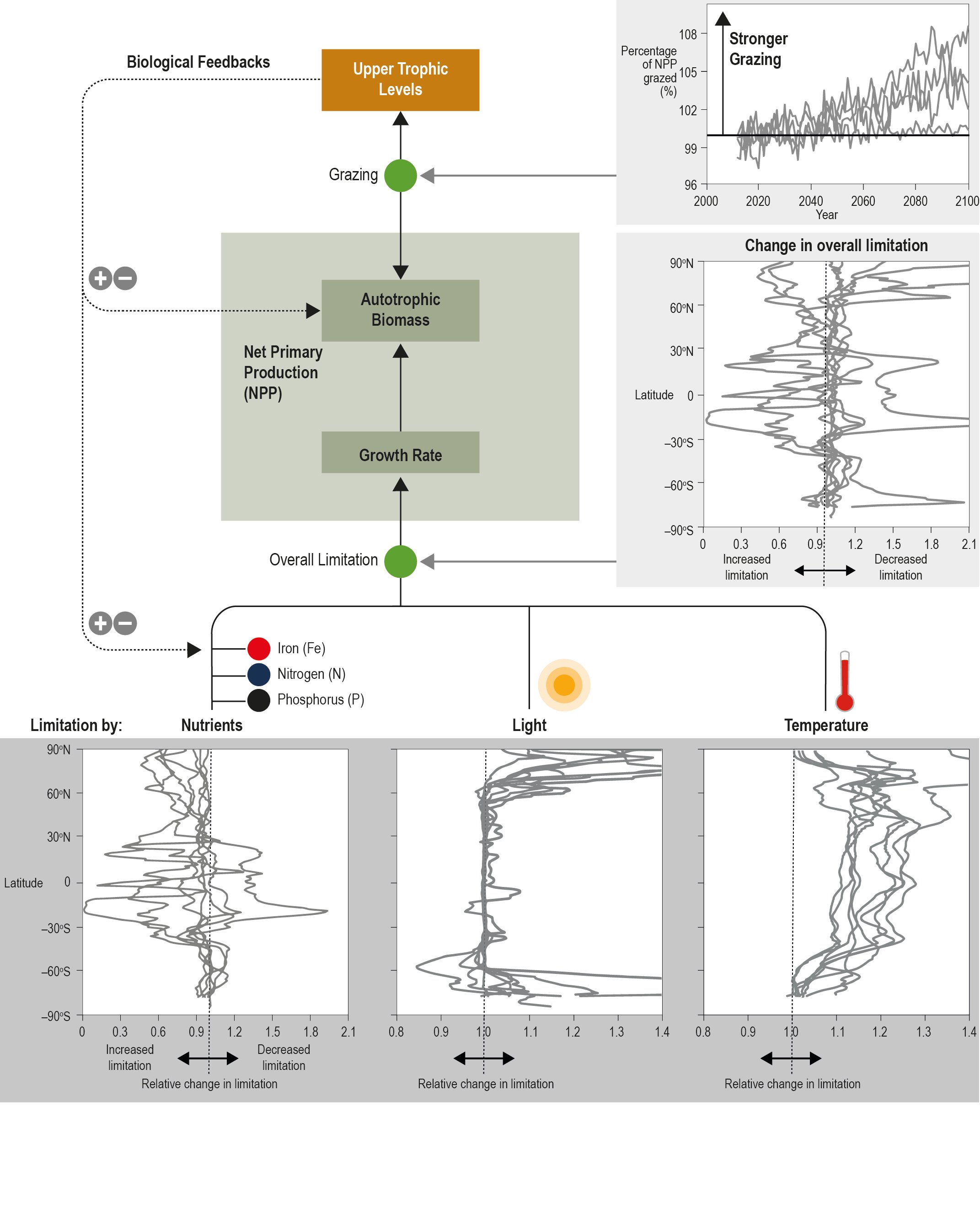
In response to ocean warming and increased stratification, open ocean nutrient cycles are being perturbed and there is high confidence that this is having a regionally variable impact on primary producers. There is currently low confidence in appraising past open ocean productivity trends, including those determined by satellites, due to newly identified region-specific drivers of microbial growth and the lack of corroborating in situ time series datasets. {5.2.2.5, 5.2.2.6}
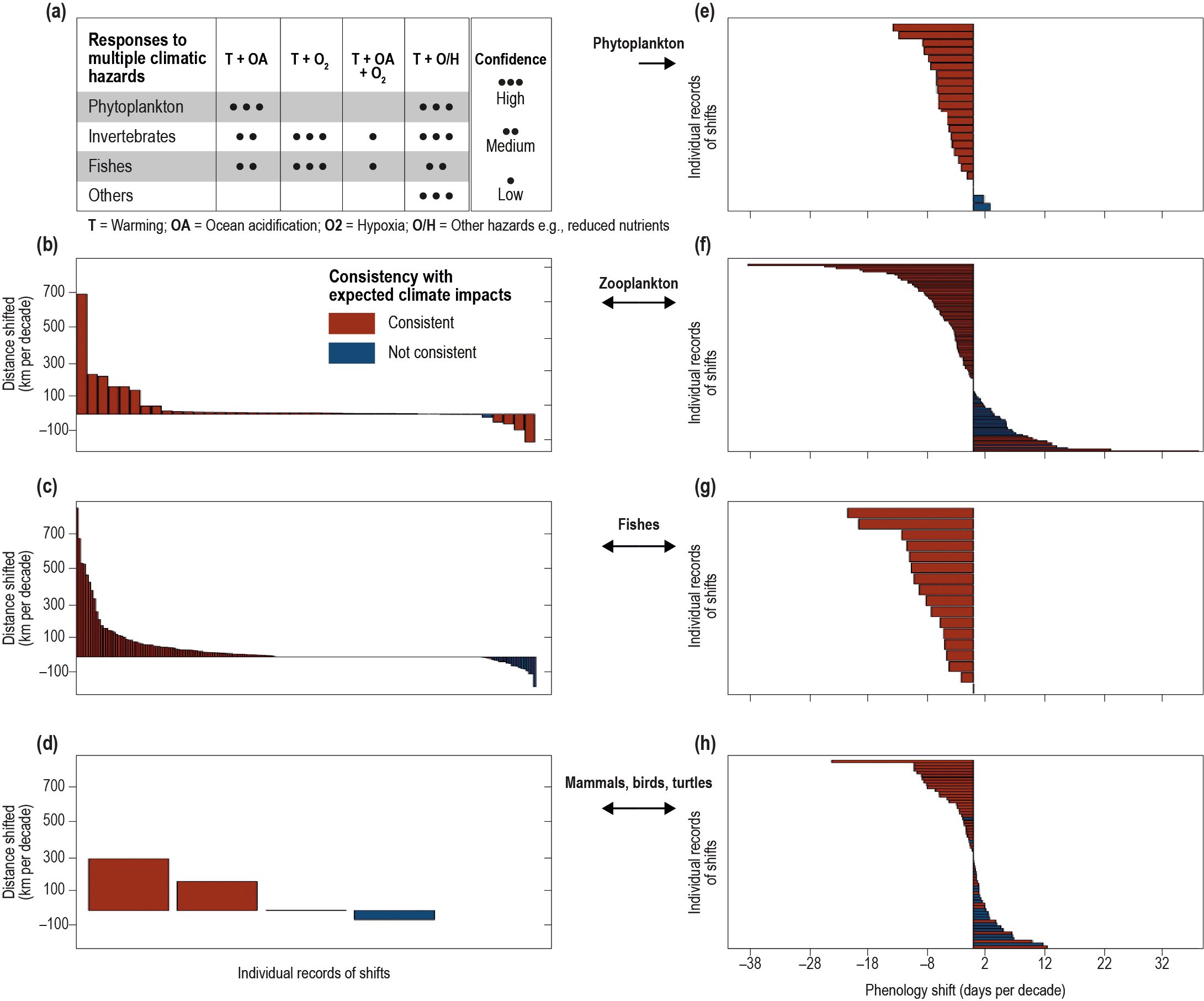
Ocean warming has contributed to observed changes in biogeography of organisms ranging from phytoplankton to marine mammals (high confidence), consequently changing community composition (high confidence), and in some cases, altering interactions between organisms (medium confidence). Observed rate of range shifts since the 1950s and its very likely range are estimated to be 51.5 ± 33.3 km per decade and 29.0 ± 15.5 km per decade for organisms in the epipelagic and seafloor ecosystems, respectively. The direction of the majority of the shifts of epipelagic organisms are consistent with a response to warming (high confidence). {5.2.3, 5.3}
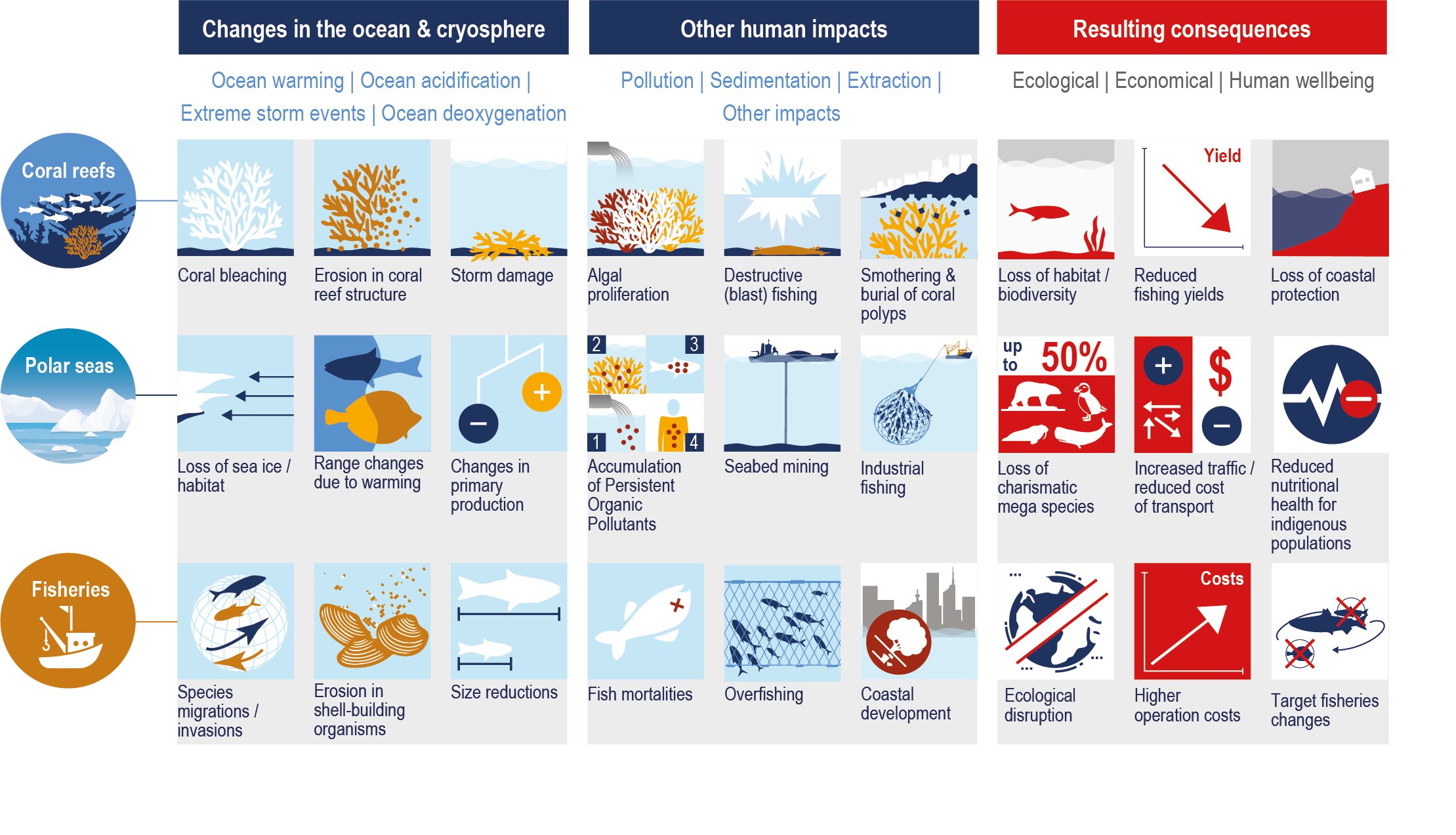
Warming-induced range expansion of tropical species to higher latitudes has led to increased grazing on some coral reefs, rocky reefs, seagrass meadows and epipelagic ecosystems, leading to altered ecosystem structure (medium confidence). Warming, sea level rise (SLR) and enhanced loads of nutrients and sediments in deltas have contributed to salinisation and deoxygenation in estuaries (high confidence), and have caused upstream redistribution of benthic and pelagic species according to their tolerance limits (medium confidence). {5.3.4, 5.3.5, 5.3.6, 5.2.3}
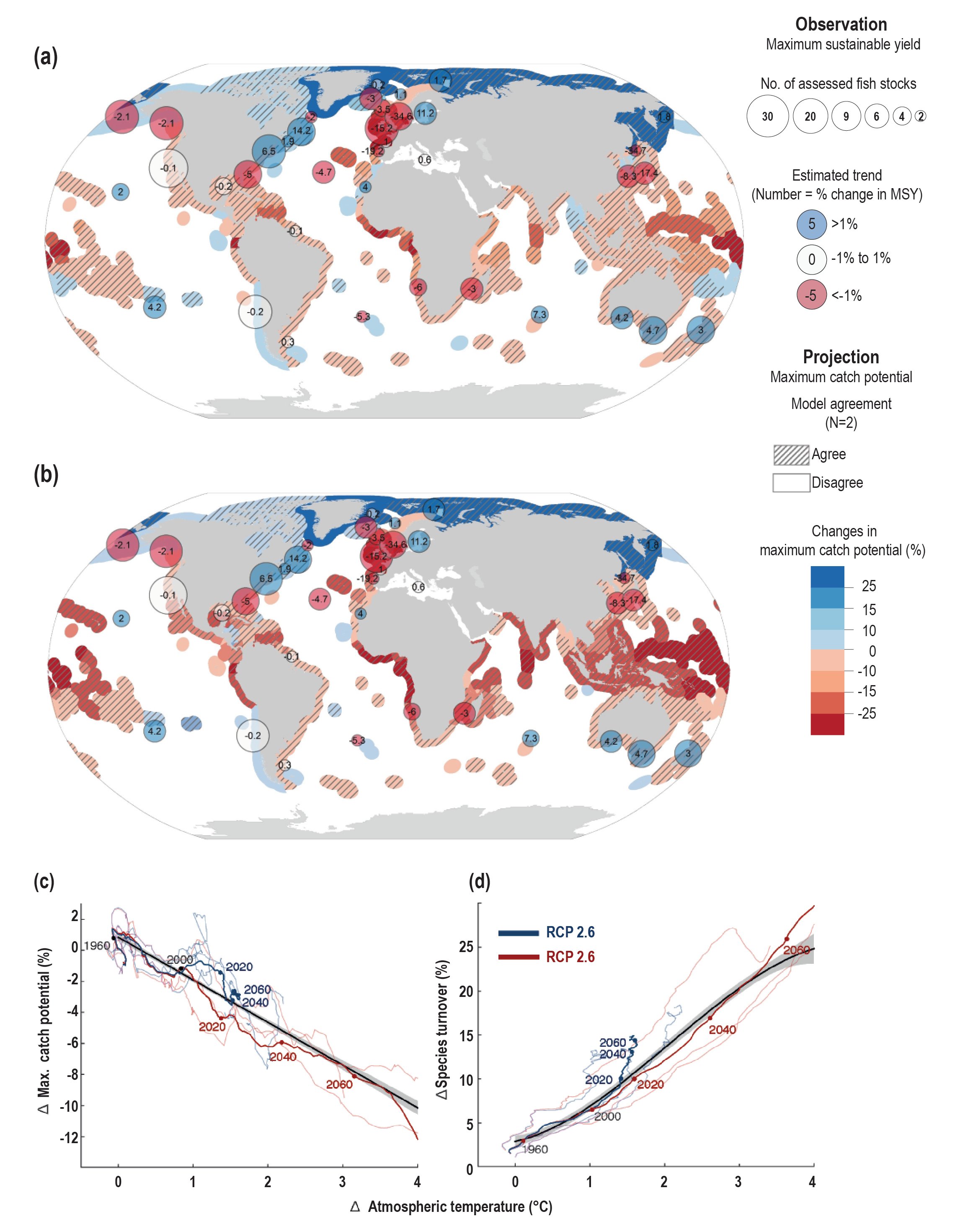
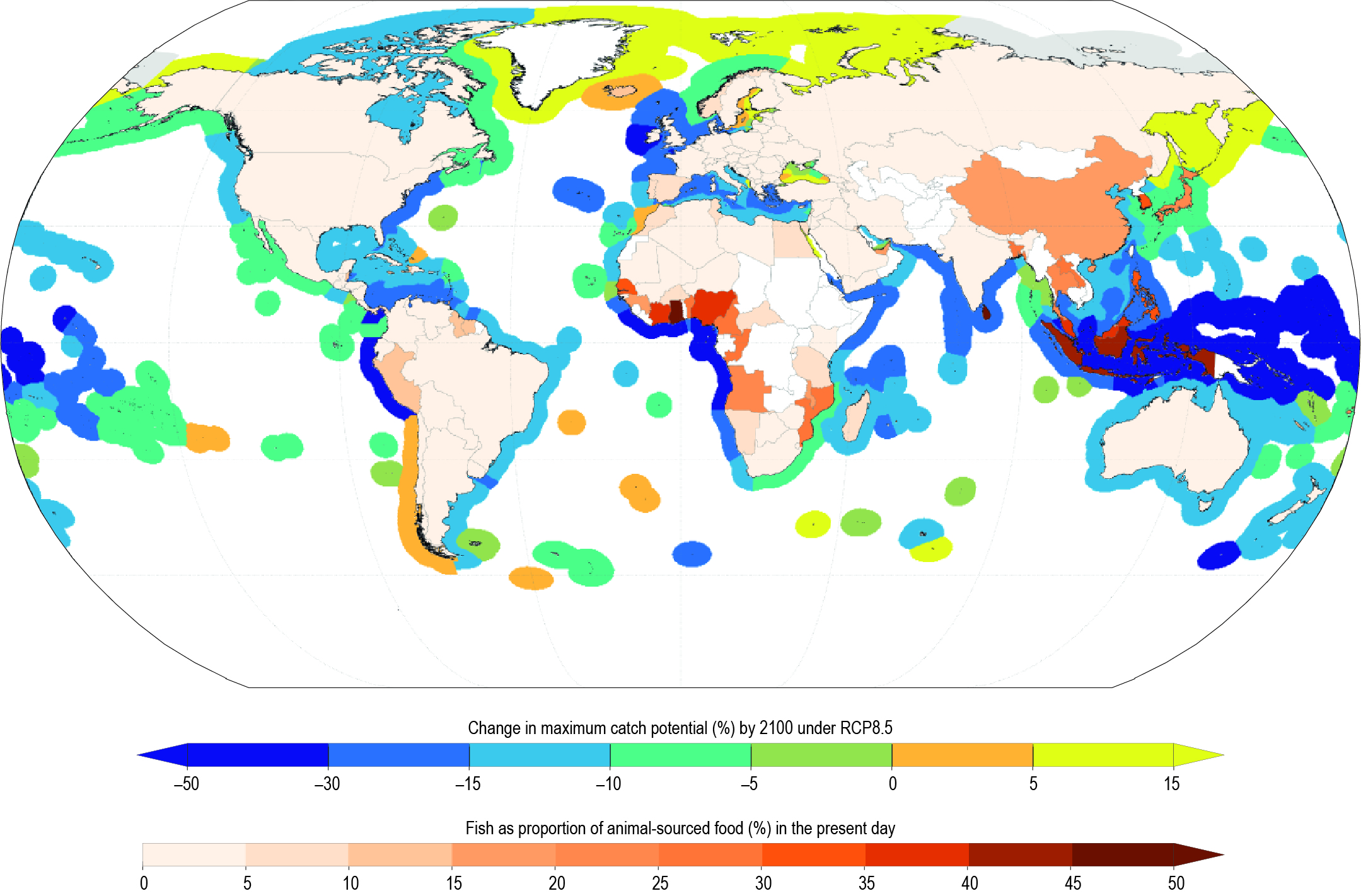
Fisheries catches and their composition in many regions are already impacted by the effects of warming and changing primary production on growth, reproduction and survival of fish stocks (high confidence). Ocean warming and changes in primary production since the 20th century are related to changes in productivity of many fish stocks (high confidence), with an average decrease of approximately 3% per decade in population replenishment and 4.1% (very likely range of 9.0% decline to 0.3% increase) in maximum catch potential (robust evidence, low agreement between fish stocks, medium confidence). Species composition of fisheries catches since the 1970s in many shelf seas ecosystems of the world is increasingly dominated by warm water species (medium confidence). {5.2.3, 5.4.1}
Warming-induced changes in spatial distribution and abundance of fish stocks have already challenged the management of some important fisheries and their economic benefits (high confidence). For existing international and national ocean and fisheries governance, there are concerns about the reduced effectiveness to achieve mandated ecological, economic, and social objectives because of observed climate impacts on fisheries resources (high confidence). {5.4.2, 5.5.2}
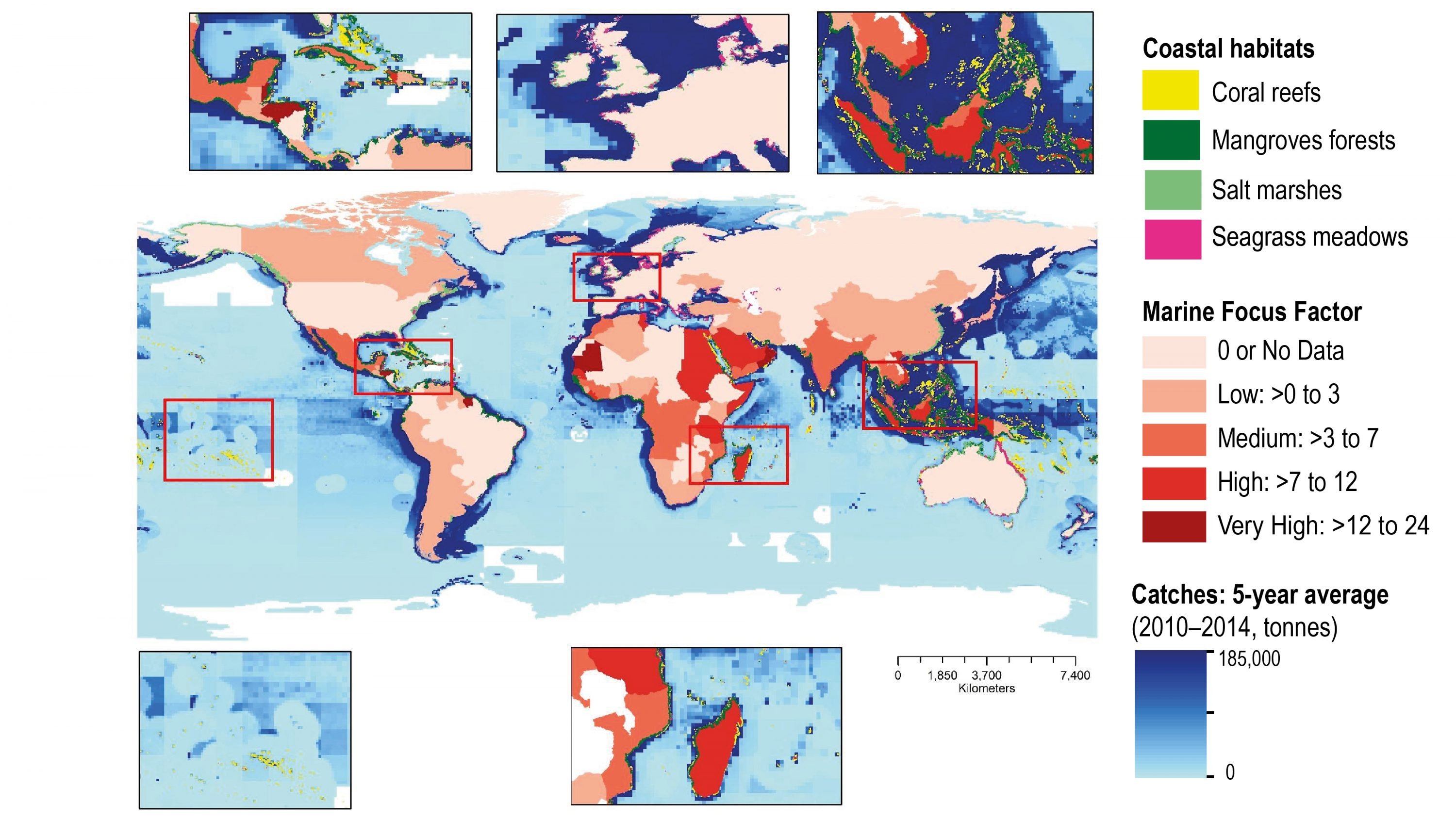
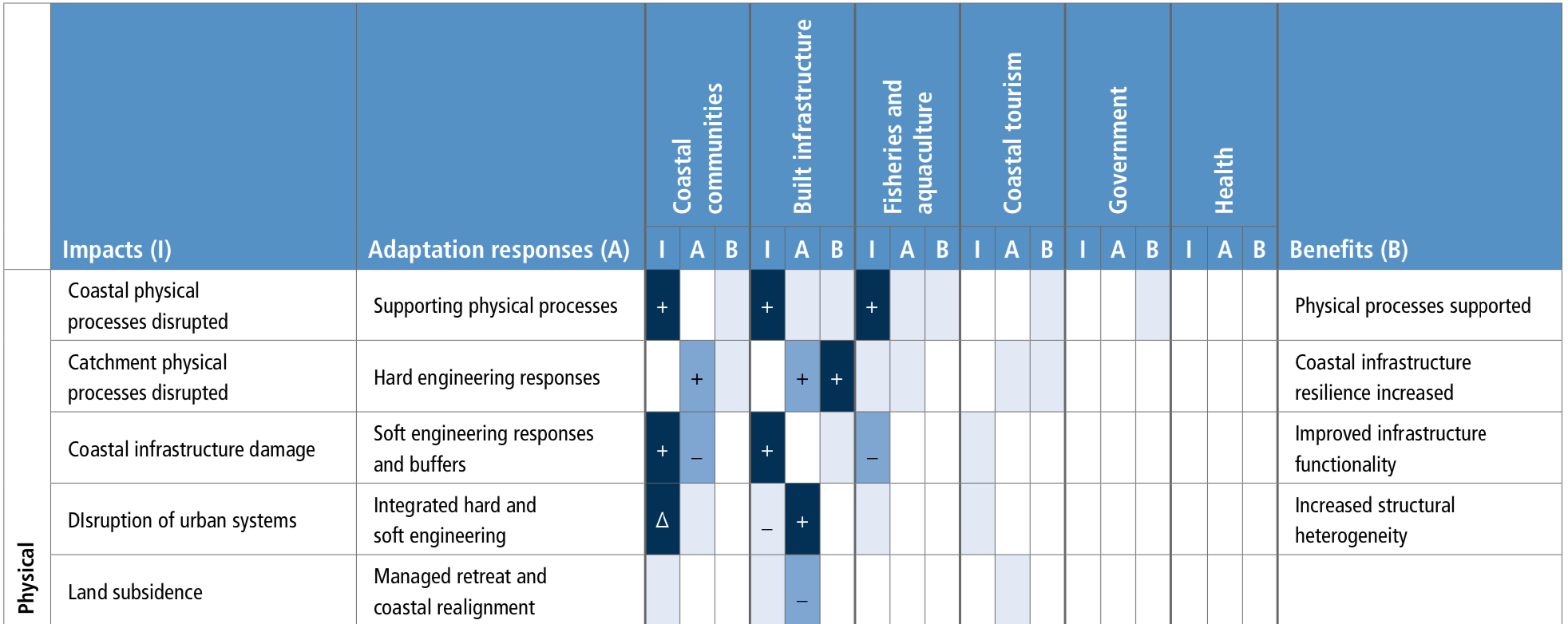
Coastal ecosystems are observed to be under stress from ocean warming and SLR that are exacerbated by non-climatic pressures from human activities on ocean and land (high confidence). Global wetland area has declined by nearly 50% relative to pre-industrial level as a result of warming, SLR, extreme climate events and other human impacts (medium confidence). Warming related mangrove encroachment into subtropical salt marshes has been observed in the past 50 years (high confidence). Distributions of seagrass meadows and kelp forests are contracting at low-latitudes that is attributable to warming (high confidence), and in some areas a loss of 36–43% following heat waves (medium confidence). Inundation, coastline erosion and salinisation are causing inland shifts in plant species distributions, which has been accelerating in the last decades (medium confidence). Warming has increased the frequency of large-scale coral bleaching events, causing worldwide reef degradation since 1997–1998 with cases of shifts to algal-dominated reefs (high confidence). Sessile calcified organisms (e.g., barnacles and mussels) in intertidal rocky shores are highly sensitive to extreme temperature events and acidification (high confidence), a reduction in their biodiversity and abundance have been observed in naturally-acidified rocky reef ecosystems (medium confidence). Increased nutrient and organic matter loads in estuaries since the 1970s have exacerbated the effects of warming on bacterial respiration and eutrophication, leading to expansion of hypoxic areas (high confidence). {5.3.1, 5.3.2, 5.3.4, 5.3.6}
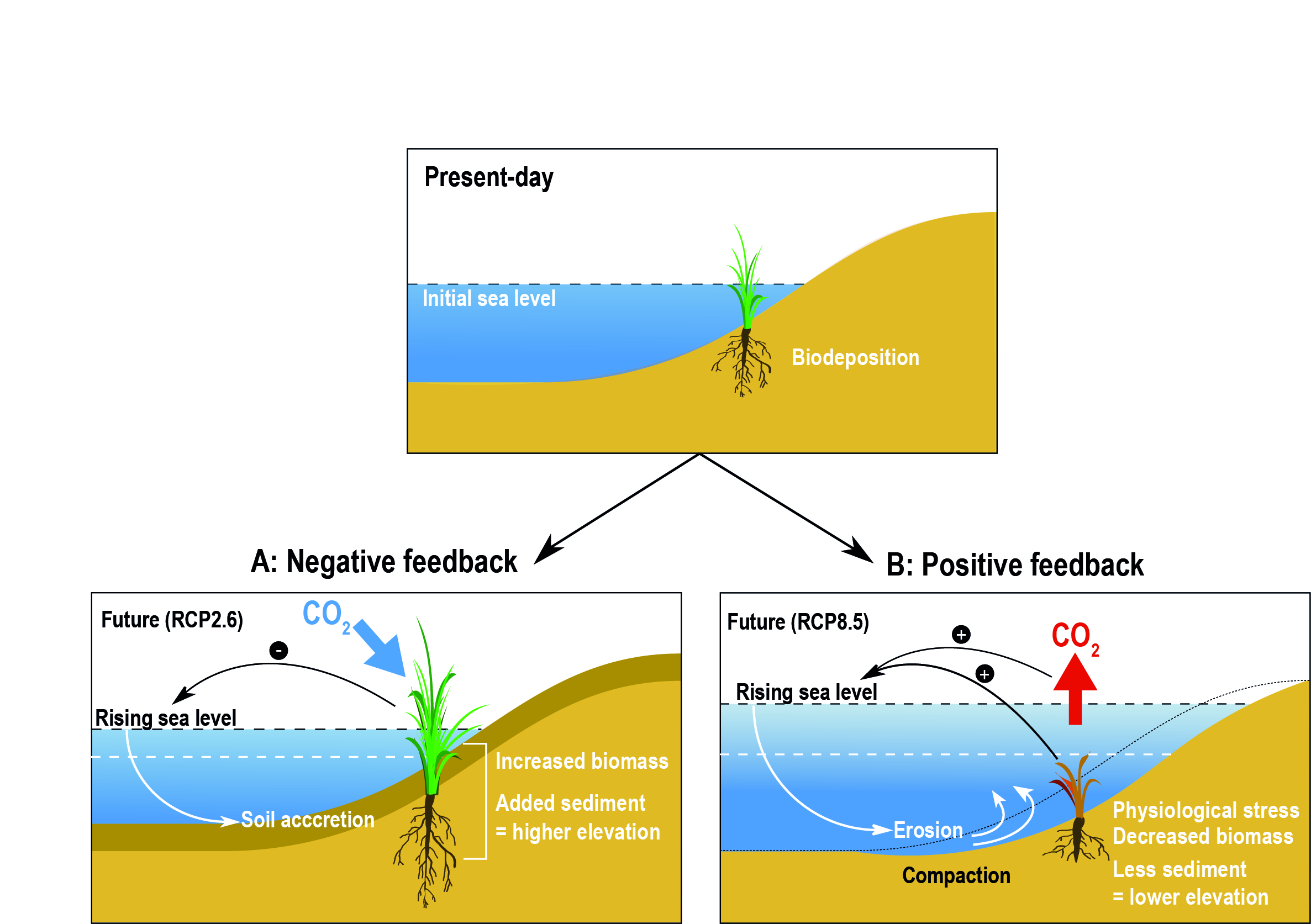
Coastal and near-shore ecosystems including salt marshes, mangrove and vegetated dunes in sandy beaches have a varying capacity to build vertically and expand laterally in response to SLR. These ecosystems provide important services including coastal protection, carbon sequestration and habitat for diverse biota (high confidence). The carbon emission associated with the loss of vegetated coastal ecosystems is estimated to be 0.04–1.46 Gt C yr-1 (high confidence). The natural capacity of ecosystems to adapt to climate impacts may be limited by human activities that fragment wetland habitats and restrict landward migration (high confidence). {5.3.2, 5.3.3, 5.4.1, 5.5.1}
Three out of the four major Eastern Boundary Upwelling Systems (EBUS) have shown large-scale wind intensification in the past 60 years (high confidence). However, the interaction of coastal warming and local winds may have affected upwelling strength, with the direction of changes varies between and within EBUS (low confidence). Increasing trends in ocean acidification in the California Current EBUS and deoxygenation in California Current and Humboldt Current EBUS are observed in the last few decades (high confidence), although there is low confidence to distinguish anthropogenic forcing from internal climate variability. The expanding California EBUS OMZ has altered ecosystem structure and fisheries catches (medium confidence). {Box 5.3}
Since the early 1980s, the occurrence of harmful algal blooms (HABs) and pathogenic organisms (e.g., Vibrio) has increased in coastal areas in response to warming, deoxygenation and eutrophication, with negative impacts on food provisioning, tourism, the economy and human health (high confidence). These impacts depend on species-specific responses to the interactive effects of climate change and other human drivers (e.g., pollution). Human communities in poorly monitored areas are among the most vulnerable to these biological hazards (medium confidence). {Box 5.4, 5.4.2}
Many frameworks for climate resilient coastal adaptation have been developed since AR5, with substantial variations in approach between and within countries, and across development status (high confidence). Few studies have assessed the success of implementing these frameworks due to the time-lag between implementation, monitoring, evaluation and reporting (medium confidence). {5.5.2}
Projections: scenarios and time horizons
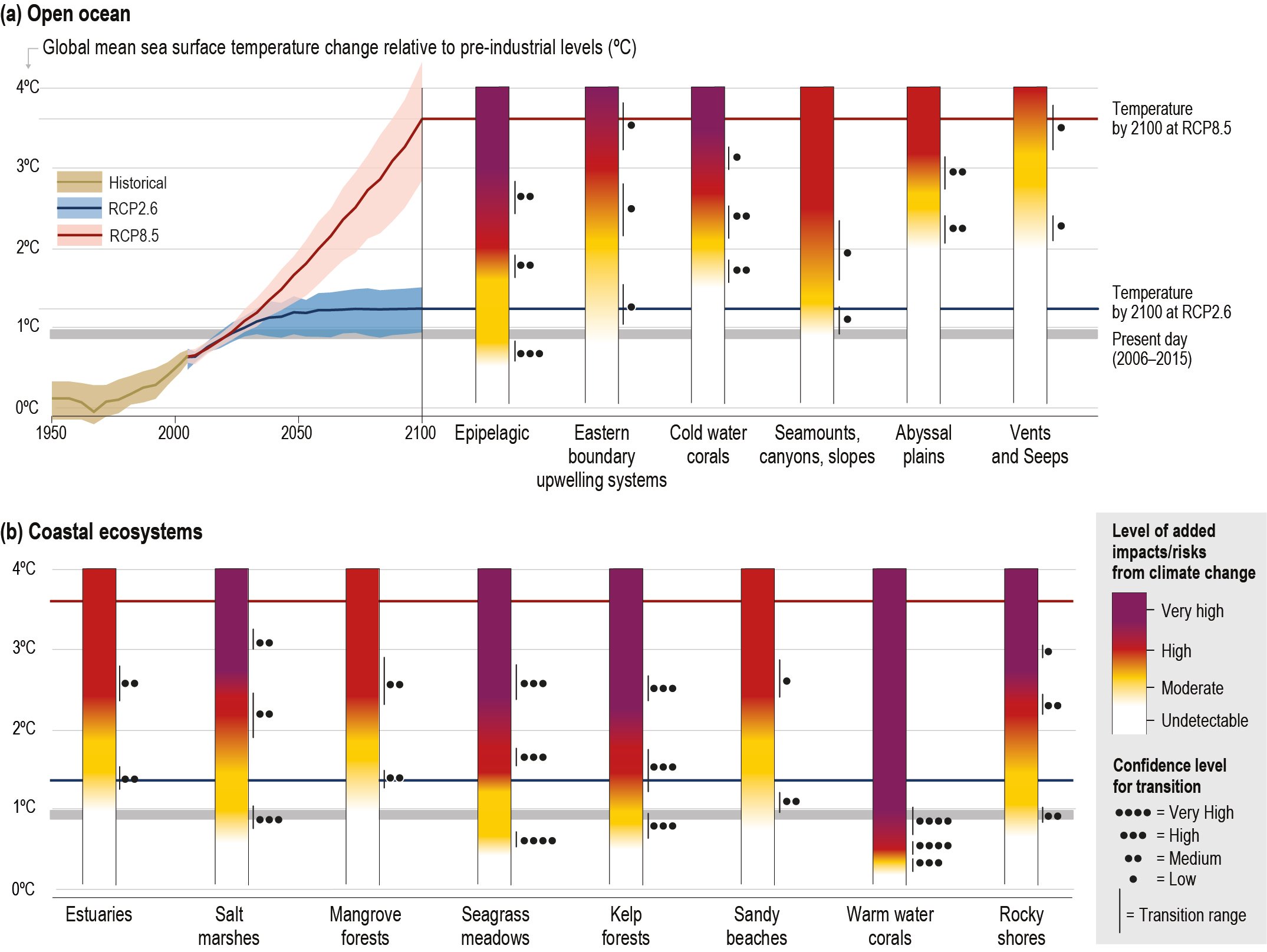
Climate models project significant changes in the ocean state over the coming century. Under the high emissions scenario (Representative Concentration Pathway (RCP)8.5) the impacts by 2090 are substantially larger and more widespread than for the low emissions scenario (RCP2.6) throughout the surface and deep ocean, including: warming (virtually certain); ocean acidification (virtually certain); decreased stability of mineral forms of calcite (virtually certain); oxygen loss (very likely); reduced near-surface nutrients (likely as not); decreased net primary productivity (high confidence); reduced fish production (likely) and loss of key ecosystems services (medium confidence) that are important for human well-being and sustainable development. {5.2.2, Box 5.1, 5.2.3, 5.2.4, 5.4}
By 2100 the ocean is very likely to warm by 2 to 4 times as much for low emissions (RCP2.6) and 5 to 7 times as much for the high emissions scenario (RCP8.5) compared with the observed changes since 1970. The 0–2000 m layer of the ocean is projected to warm by a further 2150 ZJ (very likely range 1710–2790 ZJ) between 2017 and 2100 for the RCP8.5 scenario. The 0–2000 m layer is projected to warm by 900 ZJ (very likely range 650–1340 ZJ) by 2100 for the RCP2.6 scenario, and the overall warming of the ocean will continue this century even after radiative forcing and mean surface temperatures stabilise (high confidence). {5.2.2.2}
The upper ocean will continue to stratify. By the end of the century the annual mean stratification of the top 200 m (averaged between 60°S–60°N relative to the 1986–2005 period) is projected to increase in the very likely range of 1–9% and 12–30% for RCP2.6 and RCP8.5 respectively. {5.2.2.2}
It is very likely that the majority of coastal regions will experience statistically significant changes in tidal amplitudes over the course of the 21st century. The sign and amplitude of local changes to tides are very likely to be impacted by both human coastal adaptation measures and climate drivers. {5.2.2.2.3}
It is virtually certain that surface ocean pH will decline, by 0.036–0.042 or 0.287–0.29 pH units by 2081–2100, relative to 2006–2015, for the RCP2.6 or RCP8.5 scenarios, respectively. These pH changes are very likely to cause the Arctic and Southern Oceans, as well as the North Pacific and Northwestern Atlantic Oceans to become corrosive for the major mineral forms of calcium carbonate under RCP8.5, but these changes are virtually certain to be avoided under the RCP2.6 scenario. There is increasing evidence of an increase in the seasonal exposure to acidified conditions in the future (high confidence), with a very likely increase in the amplitude of seasonal cycle of hydrogen iron concentrations of 71–90% by 2100, relative to 2000 for the RCP8.5 scenario, especially at high latitudes. {5.2.2.3}
Oxygen is projected to decline further. Globally, the oxygen content of the ocean is very likely to decline by 3.2–3.7% by 2081–2100, relative to 2006–2015, for the RCP8.5 scenario or by 1.6–2.0% for the RCP2.6 scenario. The volume of the oceans OMZ is projected to grow by a very likely range of 7.0 ± 5.6% by 2100 during the RCP8.5 scenario, relative to 1850–1900. The climate signal of oxygen loss will very likely emerge from the historical climate by 2050 with a very likely range of 59–80% of ocean area being affected by 2031–2050 and rising with a very likely range of 79–91% by 2081–2100 (RCP8.5). The emergence of oxygen loss is very likely smaller in area for the RCP2.6 scenario in the 21st century and by 2090 the emerged area is declining. {5.2.2.4, Box 5.1 Figure 1}
Overall, nitrate concentrations in the upper 100 m are very likely to decline by 9–14% across CMIP5 models by 2081–2100, relative to 2006–2015, in response to increased stratification for RCP8.5, with medium confidence in these projections due to the limited evidence of past changes that can be robustly understood and reproduced by models. There is low confidence regarding projected increases in surface ocean iron levels due to systemic uncertainties in these models. {5.2.2.5}
Climate models project that net primary productivity will very likely decline by 4–11% for RCP8.5 by 2081–2100, relative to 2006–2015. The decline is due to the combined effects of warming, stratification, light, nutrients and predation and will show regional variations between low and high latitudes (low confidence). The tropical ocean NPP will very likely decline by 7–16% for RCP8.5, with medium confidence as there are improved constraints from historical variability in this region. Globally, the sinking flux of organic matter from the upper ocean into the ocean interior is very likely to decrease by 9–16% for RCP8.5 in response to increased stratification and reduced nutrient supply, especially in tropical regions (medium confidence), which will reduce organic carbon supply to deep sea ecosystems (high confidence). The reduction in food supply to the deep sea is projected to lead to a 5–6% reduction in biomass of benthic biota over more than 97% of the abyssal seafloor by 2100 (medium confidence). {5.2.2.6, 5.2.4.2}
New ocean states for a broad suite of climate indices will progressively emerge over a substantial fractions of the ocean in the coming century (relative to past internal ocean variability), with Earth System Models (ESMs) showing an ordered emergence of first pH, followed by sea surface temperature (SST), interior oxygen, upper ocean nutrient levels and finally net primary production (NPP). The anthropogenic pH signal has very likely emerged for three quarters of the ocean prior to 1950, with little difference between scenarios. Oxygen changes will very likely emerge over 59–80% of the ocean area by 2031–2050 and rises to 79–91% by 2081–2100 (RCP8.5). The projected time of emergence for five primary drivers of marine ecosystem change (surface warming and acidification, oxygen loss, nitrate content and net primary production change) are all prior to 2100 for over 60% of the ocean area under RCP8.5 and over 30% under RCP2.6 (very likely). {Box 5.1, Box 5.1 Figure 1}
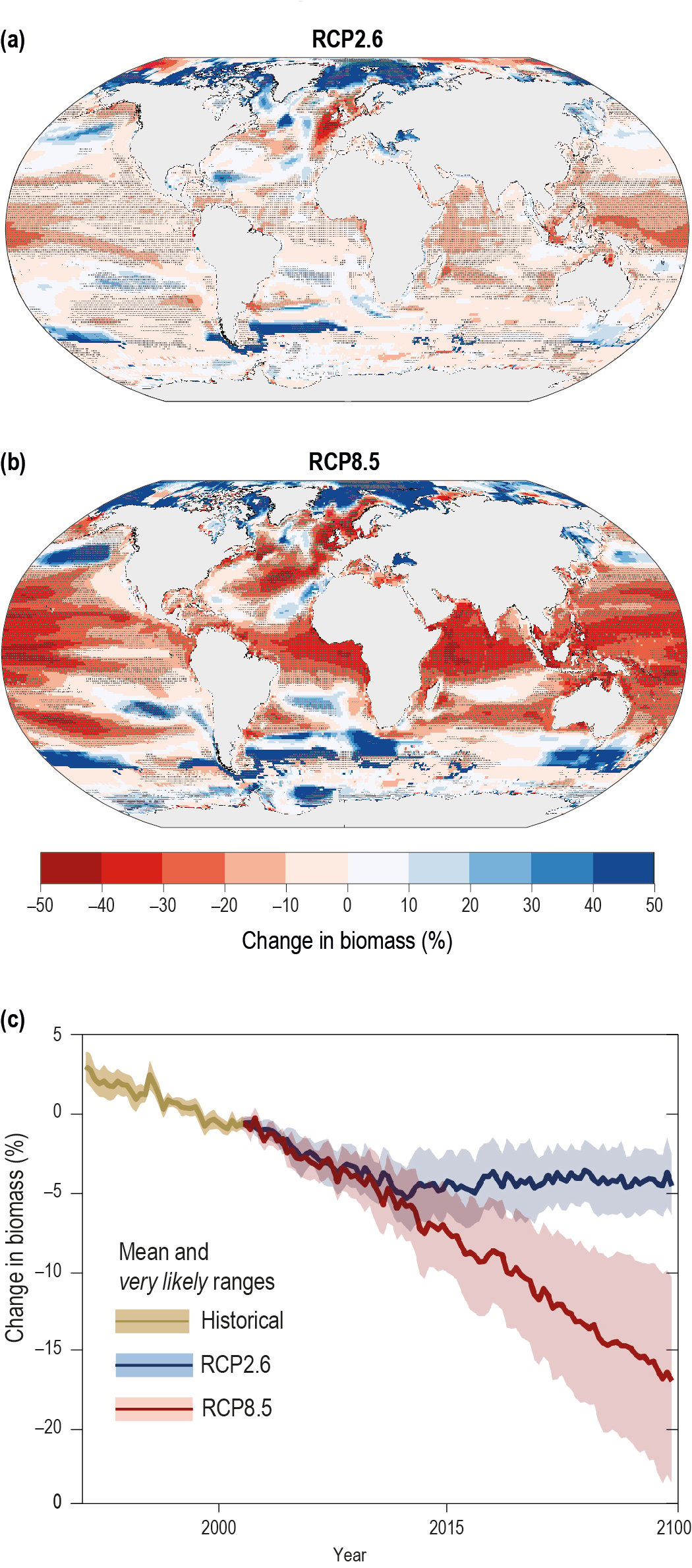
Simulated ocean warming and changes in NPP during the 21st century are projected to alter community structure of marine organisms (high confidence), reduce global marine animal biomass (medium confidence) and the maximum potential catches of fish stocks (medium confidence) with regional differences in the direction and magnitude of changes (high confidence). The global biomass of marine animals, including those that contribute to fisheries, is projected to decrease with a very likely range under RCP2.6 and RCP8.5 of 4.3 ± 2.0% and 15.0 ± 5.9%, respectively, by 2080–2099 relative to 1986–2005. The maximum catch potential is projected to decrease by 3.4% to 6.4% (RCP2.6) and 20.5% to 24.1% (RCP8.5) in the 21st century. {5.4.1}
Projected decreases in global marine animal biomass and fish catch potential could elevate the risk of impacts on income, livelihood and food security of the dependent human communities (medium confidence). Projected climate change impacts on fisheries also increase the risk of potential conflicts among fishery area users and authorities or among two different communities within the same country (medium confidence), exacerbated through competing resource exploitation from international actors and mal-adapted policies (low confidence). {5.2.3, 5.4, 5.5.3}
Projected decrease in upper ocean export of organic carbon to the deep seafloor is expected to result in a loss of animal biomass on the deep seafloor by 5.2–17.6% by 2090–2100 compared to the present (2006–2015) under RCP8.5 with regional variations (medium confidence). Some increases are projected in the polar regions, due to enhanced stratification in the surface ocean, reduced primary production and shifts towards small phytoplankton (medium confidence). The projected impacts on biomass in the abyssal seafloor are larger under RCP8.5 than RCP4.5 (very likely). The increase in climatic hazards beyond thresholds of tolerance of deep sea organisms will increase the risk of loss of biodiversity and impacts on functioning of deep water column and seafloor that is important to support ecosystem services, such as carbon sequestration (medium confidence). {5.2.4}
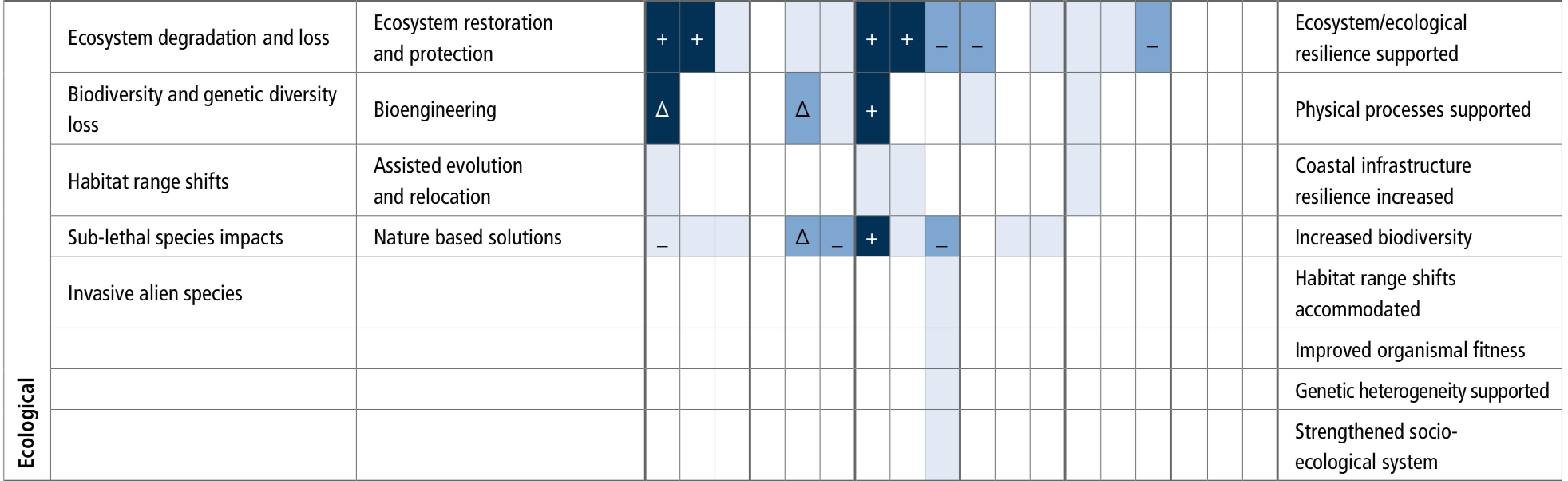
Structure and functions of all types of coastal ecosystems will continue to be at moderate to high risk under the RCP2.6 scenario (medium confidence) and will face high to very high risk under the RCP8.5 scenario (high confidence) by 2100. Seagrass meadows (high confidence) and kelp forests (high confidence) will face moderate to high risk at temperature above 1.5oC global sea surface warming. Coral reefs will face very high risk at temperatures 1.5°C of global sea surface warming (very high confidence). Intertidal rocky shores are also expected to be at very high risk (transition above 3°C) under the RCP8.5 scenario (medium confidence). These ecosystems have low to moderate adaptive capacity, as they are highly sensitive to ocean temperatures and acidification. The ecosystems with moderate to high risk (transition above 1.8°C) under future emissions scenarios are mangrove forests, sandy beaches, estuaries and salt marshes (medium confidence). Estuaries and sandy beaches are subject to highly dynamic hydrological and geomorphological processes, giving them more natural adaptive capacity to climate hazards. In these systems, sediment relocation, soil accretion and landward expansion of vegetation may initially mitigate against flooding and habitat loss, but salt marshes in particular will be at very high risk in the context of SLR and extreme climate-driven erosion under RCP8.5. {5.3, Figure 5.16}
Expected coastal ecosystem responses over the 21st century are habitat contraction, migration and loss of biodiversity and functionality. Pervasive human coastal disturbances will limit natural ecosystem adaptation to climate hazards (high confidence). Global coastal wetlands will lose between 20–90% of their area depending on emissions scenario with impacts on their contributions to carbon sequestration and coastal protection (high confidence). Kelp forests at low-latitudes and temperate seagrass meadows will continue to retreat as a result of intensified extreme temperatures, and their low dispersal ability will elevate the risk of local extinction under RCP8.5 (high confidence). Intertidal rocky shores will continue to be affected by ocean acidification, warming, and extreme heat exposure during low tide emersion, causing reduction of calcareous species and loss of ecosystem biodiversity and complexity shifting towards algae dominated habitats (high confidence). Salinisation and expansion of hypoxic conditions will intensify in eutrophic estuaries, especially in mid and high latitudes with microtidal regimes (high confidence). Sandy beach ecosystems will increasingly be at risk of eroding, reducing the habitable area for dependent organisms (high confidence). {5.3, 5.4.1}
Almost all coral reefs will degrade from their current state, even if global warming remains below 2ᴼC (very high confidence), and the remaining shallow coral reef communities will differ in species composition and diversity from present reefs (very high confidence). These declines in coral reef health will greatly diminish the services they provide to society, such as food provision (high confidence), coastal protection (high confidence) and tourism (medium confidence). {5.3.4, 5.4.1}
Multiple hazards of warming, deoxygenation, aragonite under-saturation and decrease in flux of organic carbon from the surface ocean will decrease calcification and exacerbate the bioerosion and dissolution of the non-living component of cold water coral. Habitat-forming, cold water corals will be vulnerable where temperature and oxygen exceed the species’ thresholds (medium confidence). Reduced particulate food supply is projected to be experienced by 95% of cold water coral ecosystems by 2100 under RCP8.5 relative to the present, leading to a very likely range of 8.6 ± 2% biomass loss (medium confidence). {5.2.4, Box 5.2}
Anthropogenic changes in EBUS will emerge primarily in the second half of the 21st century (medium confidence). EBUS will be impacted by climate change in different ways, with strong regional variability with consequences for fisheries, recreation and climate regulation (medium confidence). The Pacific EBUS are projected to have calcium carbonate undersaturation in surface waters within a few decades under RCP8.5 (high confidence); combined with warming and decreasing oxygen levels, this will increase the impacts on shellfish larvae, benthic invertebrates and demersal fishes (high confidence) and related fisheries and aquaculture (medium confidence). The inherent natural variability of EBUS, together with uncertainties in present and future trends in the intensity and seasonality of upwelling, coastal warming and stratification, primary production and biogeochemistry of source waters poses large challenges in projecting the response of EBUS to climate change and to the adaptation of governance of biodiversity conservation and living marine resources in EBUS (high confidence). {Box 5.3}
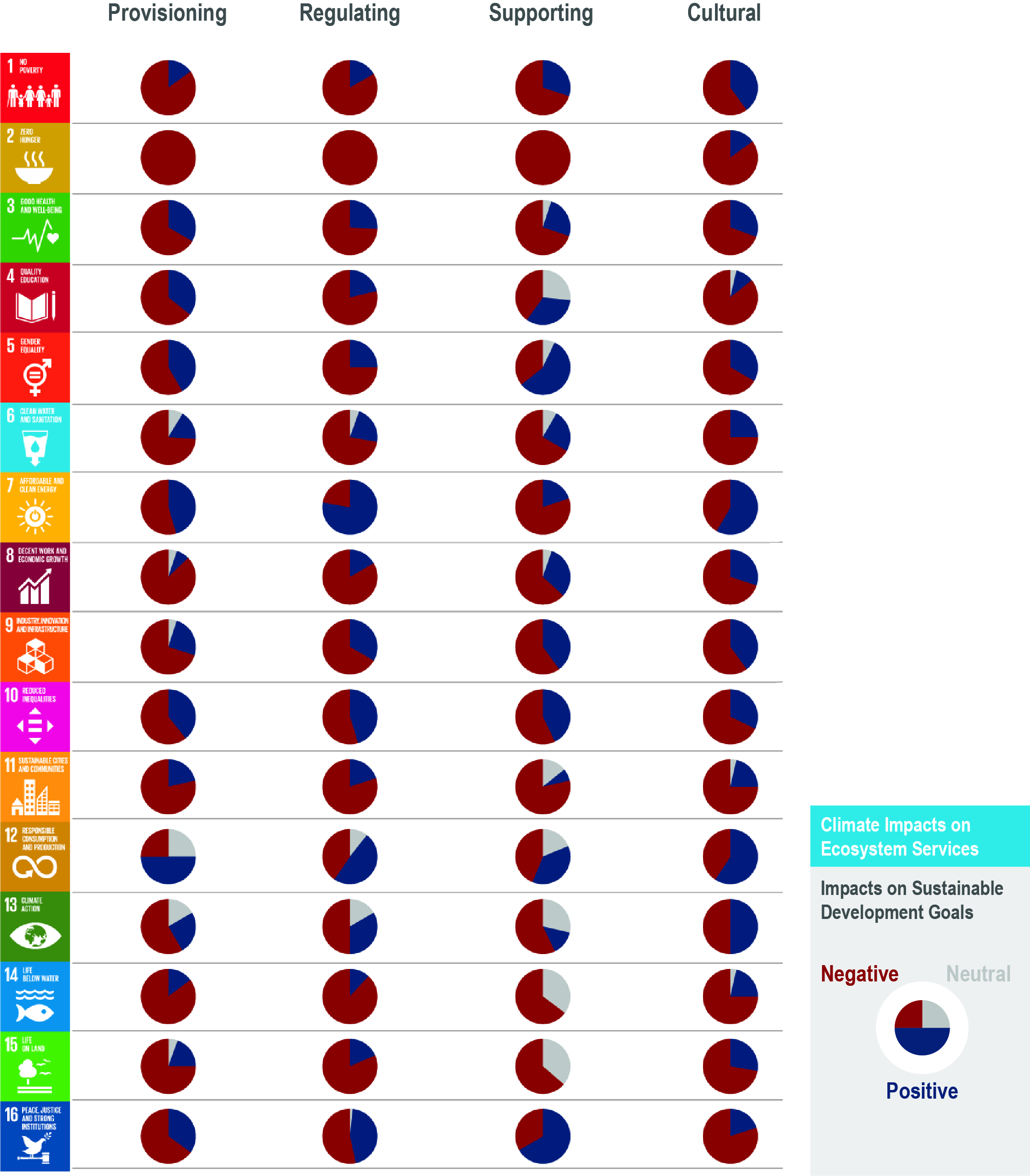
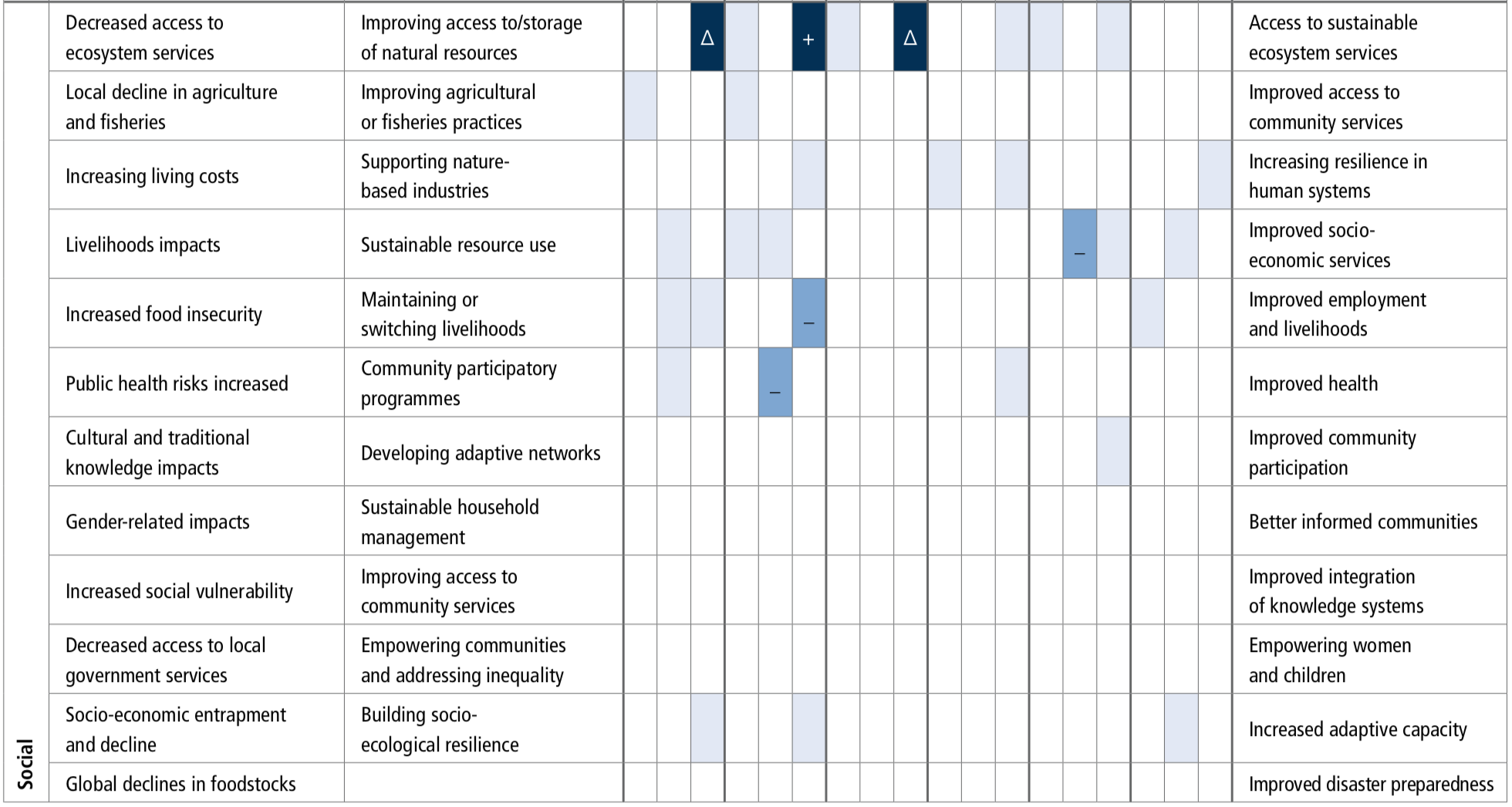
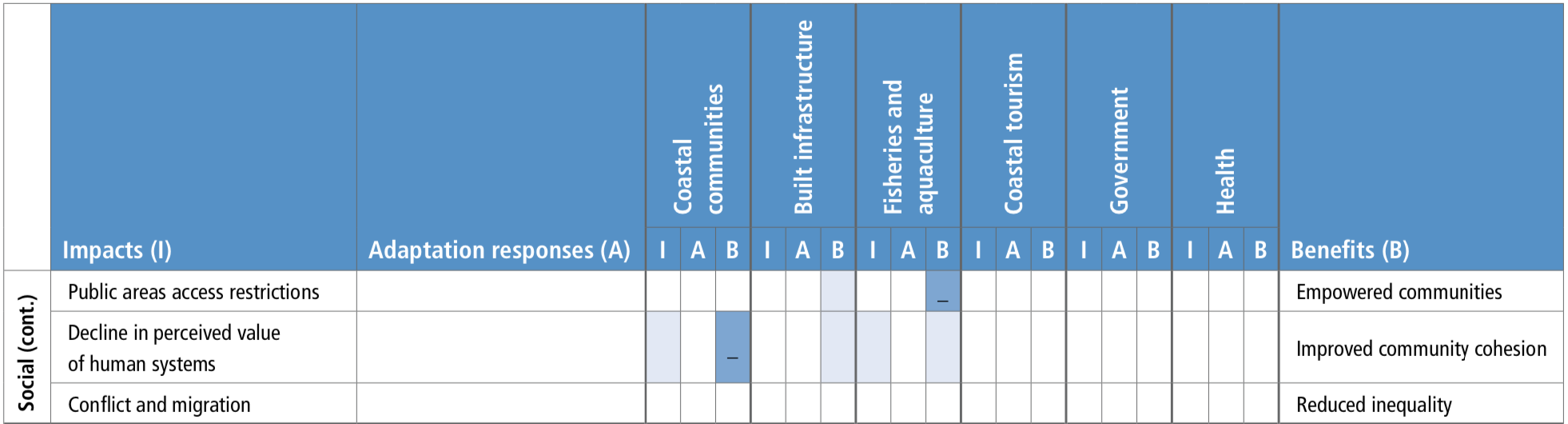
Climate change impacts on ecosystems and their goods and services threatens key cultural dimensions of lives and livelihoods. These threats include erosion of Indigenous and non-indigenous culture, their knowledge about the ocean and knowledge transmission, reduced access to traditional food, loss of opportunities for aesthetic and spiritual appreciation of the ecosystems, and marine recreational activities (medium confidence). Ultimately, these can lead to the loss of part of people’s cultural identity and values beyond the rate at which identify and values can be adjusted or substituted (medium confidence). {5.4.2}
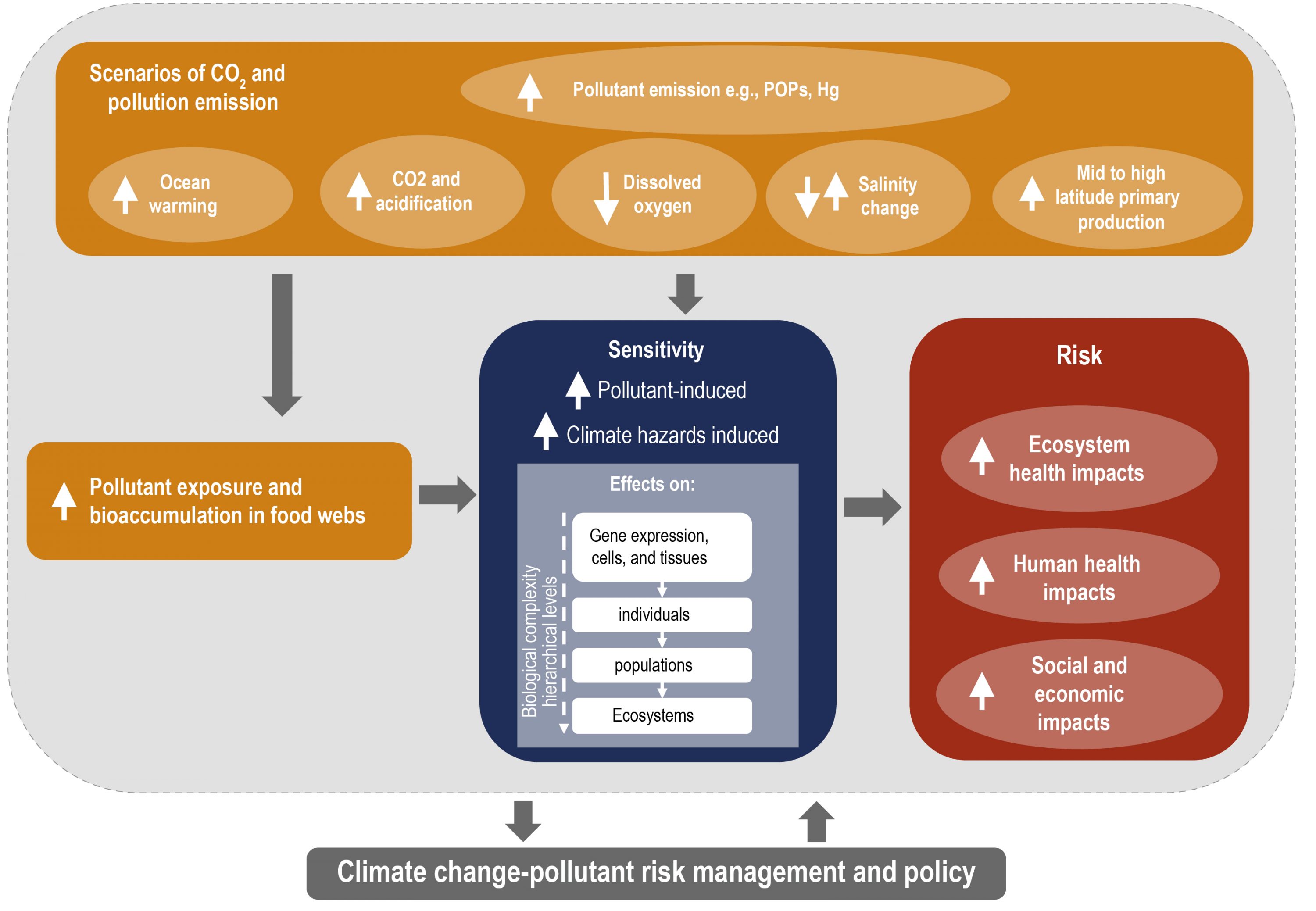
Climate change increases the exposure and bioaccumulation of contaminants such as persistent organic pollutants and mercury (medium confidence), and their risk of impacts on marine ecosystems and seafood safety (high agreement, medium evidence, medium confidence). Such risks are particularly large for top predators and for human communities that have high consumption on these organisms, including coastal Indigenous communities (medium confidence). {5.4.2}
Shifting distributions of fish stocks between governance jurisdictions will increase the risk of potential conflicts among fishery area users and authorities or different communities within the same country (medium confidence). These fishery governance related risks are widespread under high emissions scenarios with regional hotspots (medium confidence), and highlight the limits of existing natural resource management frameworks for addressing ecosystem change (high confidence). {5.2.5, 5.4.2.1.3, 5.5, 5.5.2}
Response options to enhance resilience
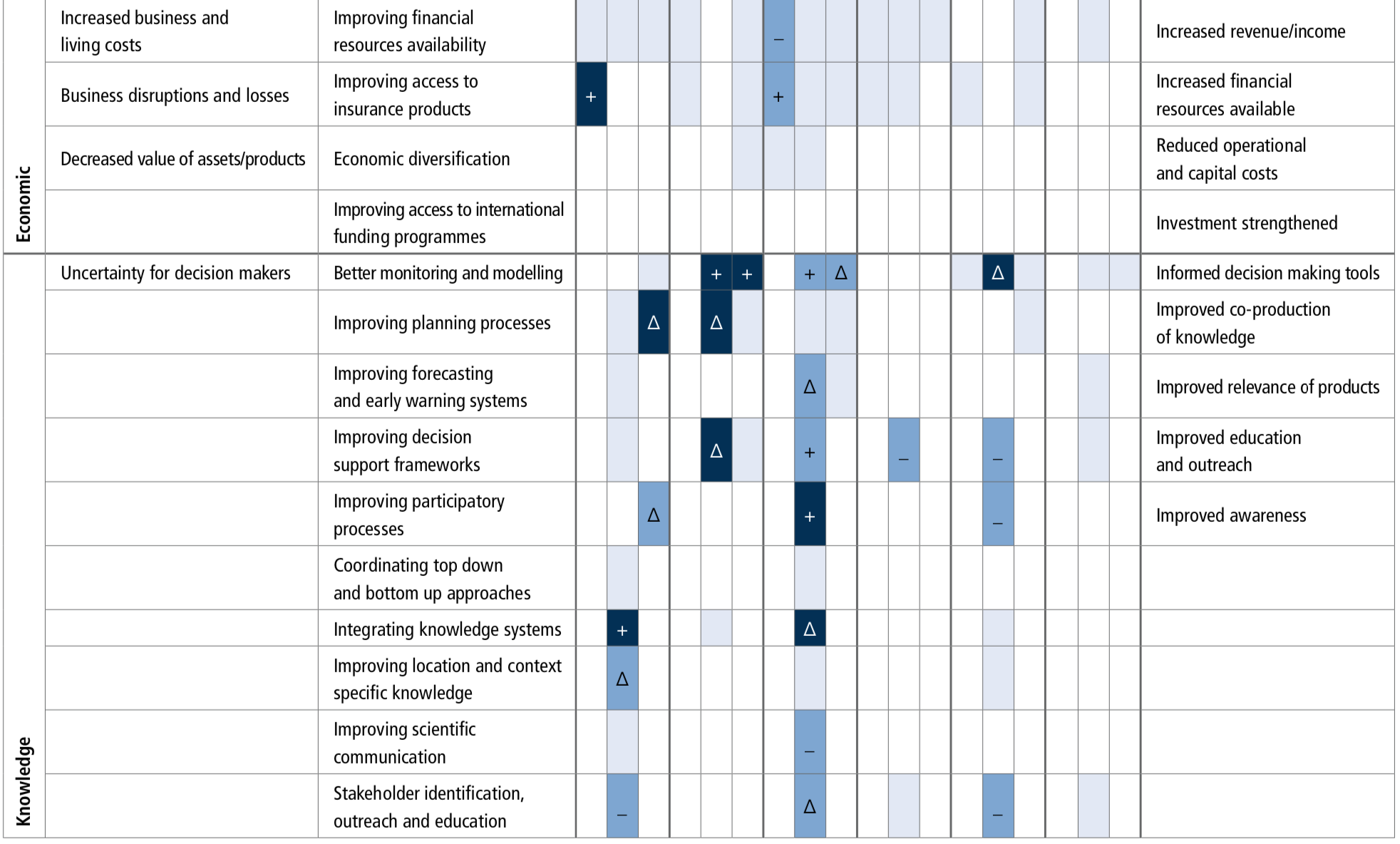
There is clear evidence for observed climate change impacts throughout the ocean with consequences for human communities and require options to reduce risks and impacts. Coastal blue carbon can contribute to mitigation for many nations but its global scope is modest (offset of <2% of current emissions) (likely). Some ocean indices are expected to emerge earlier than others (e.g., warming, acidification and effects on fish stocks) and could therefore be used to prioritise planning and building resilience. The survival of some keystone ecosystems (e.g., coral reefs) are at risk, while governance structures are not well-matched to the spatial and temporal scale of climate change impacts on ocean systems. Ecosystem restoration may be able to locally reduce climate risks (medium confidence) but at relatively high cost and effectiveness limited to low emissions scenarios and to less sensitive systems (high confidence). {5.2, 5.3, 5.4, 5.5}
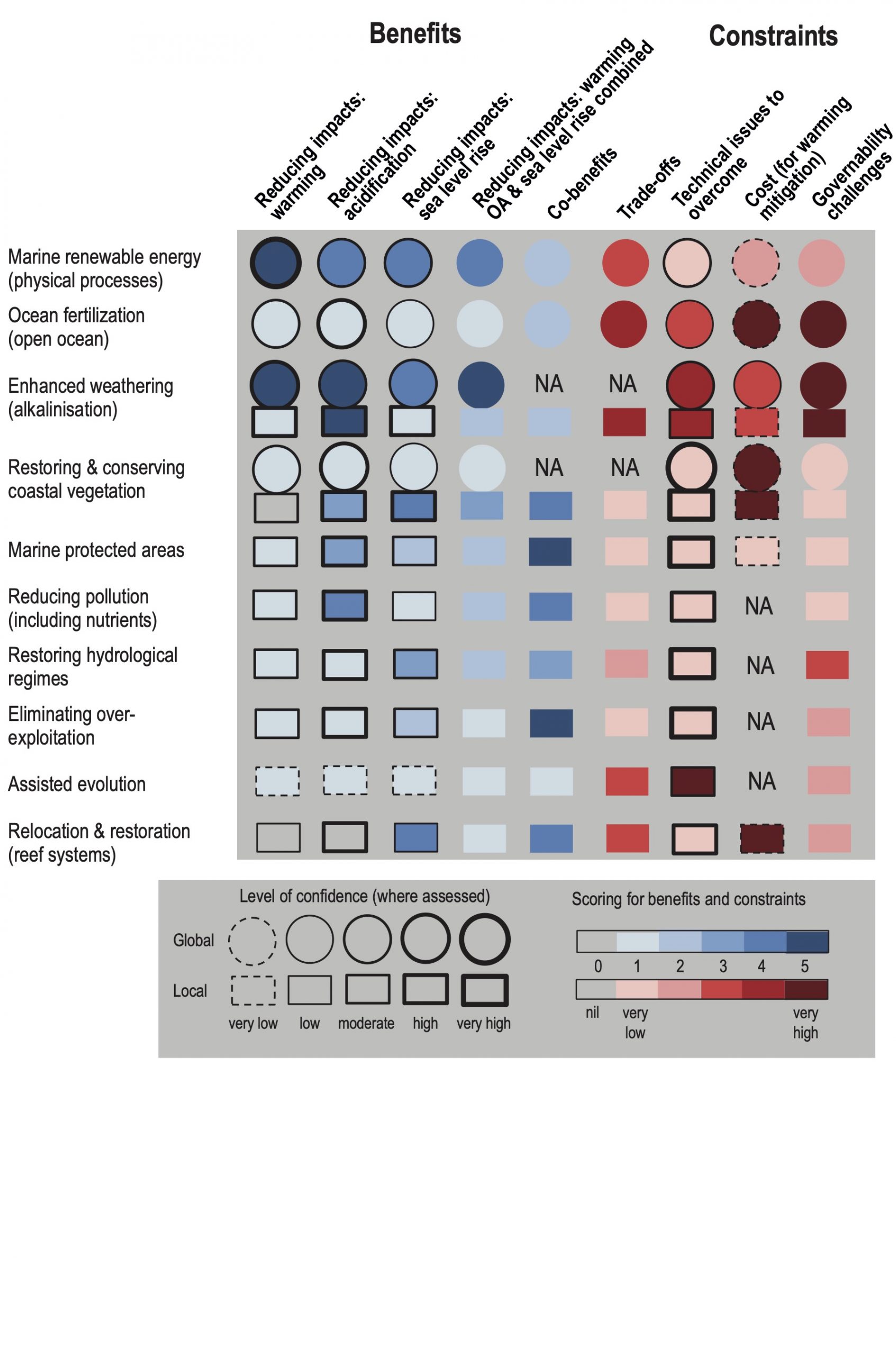
Coastal blue carbon ecosystems, such as mangroves, salt marshes and seagrasses, can help reduce the risks and impacts of climate change, with multiple co-benefits. Some 151 countries around the world contain at least one of these coastal blue carbon ecosystems and 71 countries contain all three. Below-ground carbon storage in vegetated marine habitats can be up to 1000 tC ha-1, much higher than most terrestrial ecosystems (high confidence). Successful implementation of measures to maintain and promote carbon storage in such coastal ecosystems could assist several countries in achieving a balance between emissions and removals of greenhouse gases (medium confidence). Conservation of these habitats would also sustain the wide range of ecosystem services they provide and assist with climate adaptation through improving critical habitats for biodiversity, enhancing local fisheries production, and protecting coastal communities from SLR and storm events (high confidence). The climate mitigation effectiveness of other natural carbon removal processes in coastal waters, such as seaweed ecosystems and proposed non-biological marine CO2 removal methods, are smaller or currently have higher associated uncertainties. Seaweed aquaculture warrants further research attention. {5.5.1.1, 5.5.1.1, 5.5.1, 5.5.2, 5.5.1.1.3, 5.5.1.1.4}
The potential climatic benefits of blue carbon ecosystems can only be a very modest addition to, and not a replacement for, the very rapid reduction of greenhouse gas emissions. The maximum global mitigation benefits of cost-effective coastal wetland restoration is unlikely to be more than 2% of current total emissions from all sources. Nevertheless, the protection and enhancement of coastal blue carbon can be an important contribution to both mitigation and adaptation at the national scale. The feasibility of climate mitigation by open ocean fertilisation of productivity is limited to negligible, due to the likely decadal-scale return to the atmosphere of nearly all the extra carbon removed, associated difficulties in carbon accounting, risks of unintended side effects and low acceptability. Other human interventions to enhance marine carbon uptake, for example, ocean alkalinisation (enhanced weathering), would also have governance challenges, with the increased risk of undesirable ecological consequences (high confidence). {5.5.1.2}
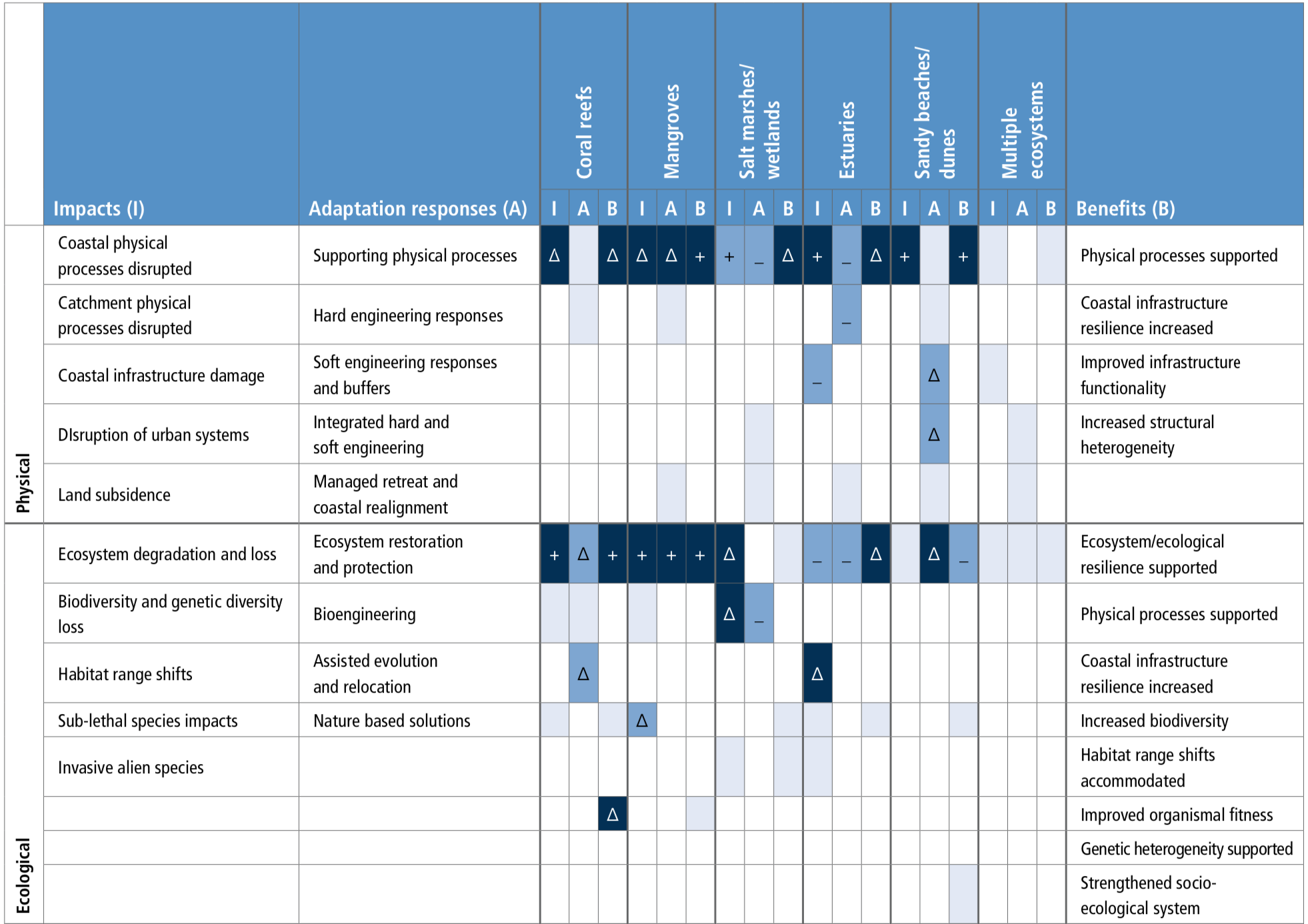
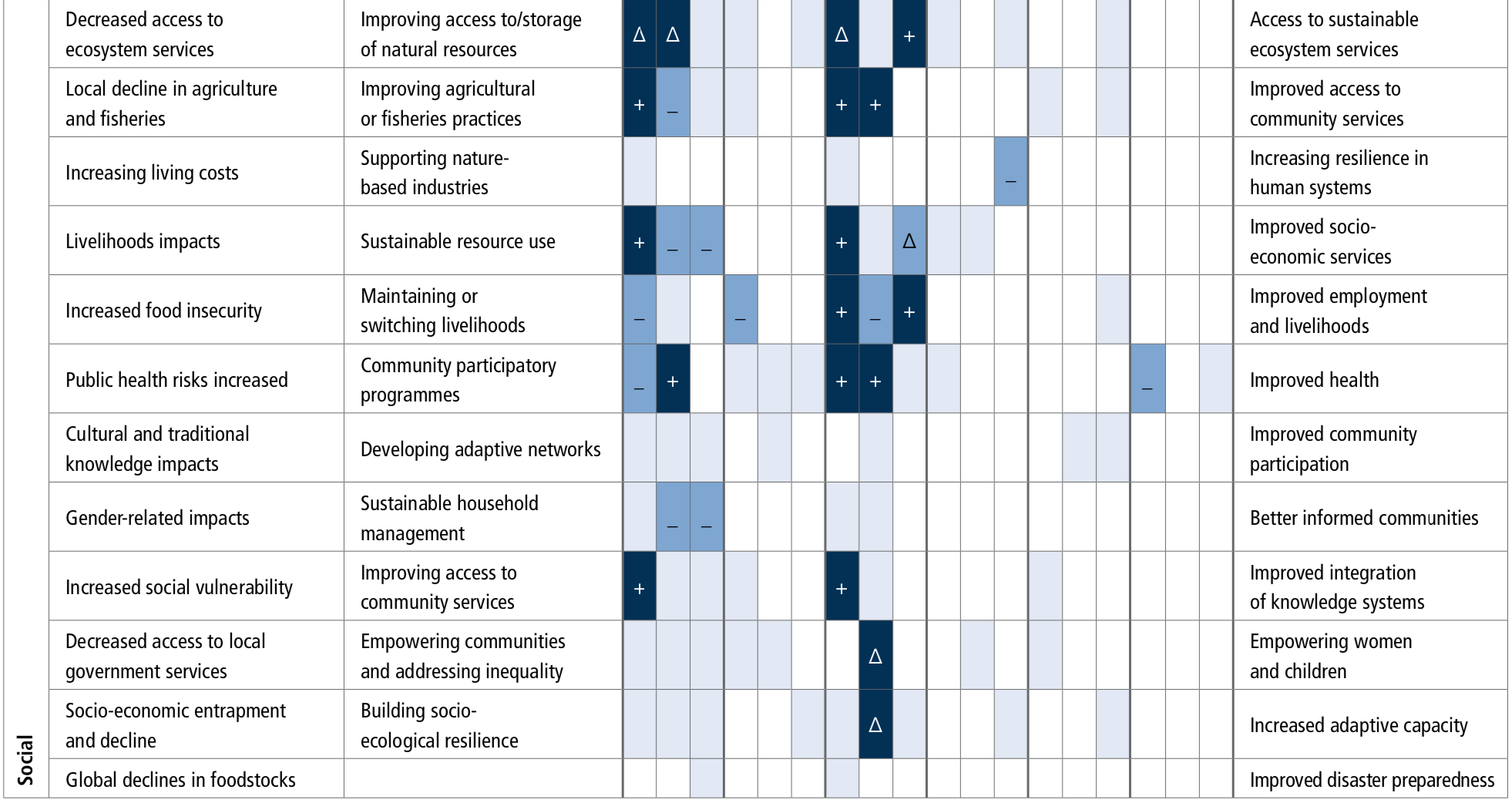
Socioinstitutional adaptation responses are more frequently reported in the literature than ecosystem-based and built infrastructure approaches. Hard engineering responses are more effective when supported by ecosystem-based adaptation approaches (high agreement), and both approaches are enhanced by combining with socioinstitutional approaches for adaptation (high confidence). Stakeholder engagement is necessary (robust evidence, high agreement). {5.5.2}
Ecosystem-based adaptation is a cost-effective coastal protection tool that can have many co-benefits, including supporting livelihoods, contributing to carbon sequestration and the provision of a range of other valuable ecosystem services (high confidence). Such adaptation does, however, assume that the climate can be stabilised. Under changing climatic conditions there are limits to the effectiveness of ecosystem-based adaptation, and these limits are currently difficult to determine. {5.5.2.1}
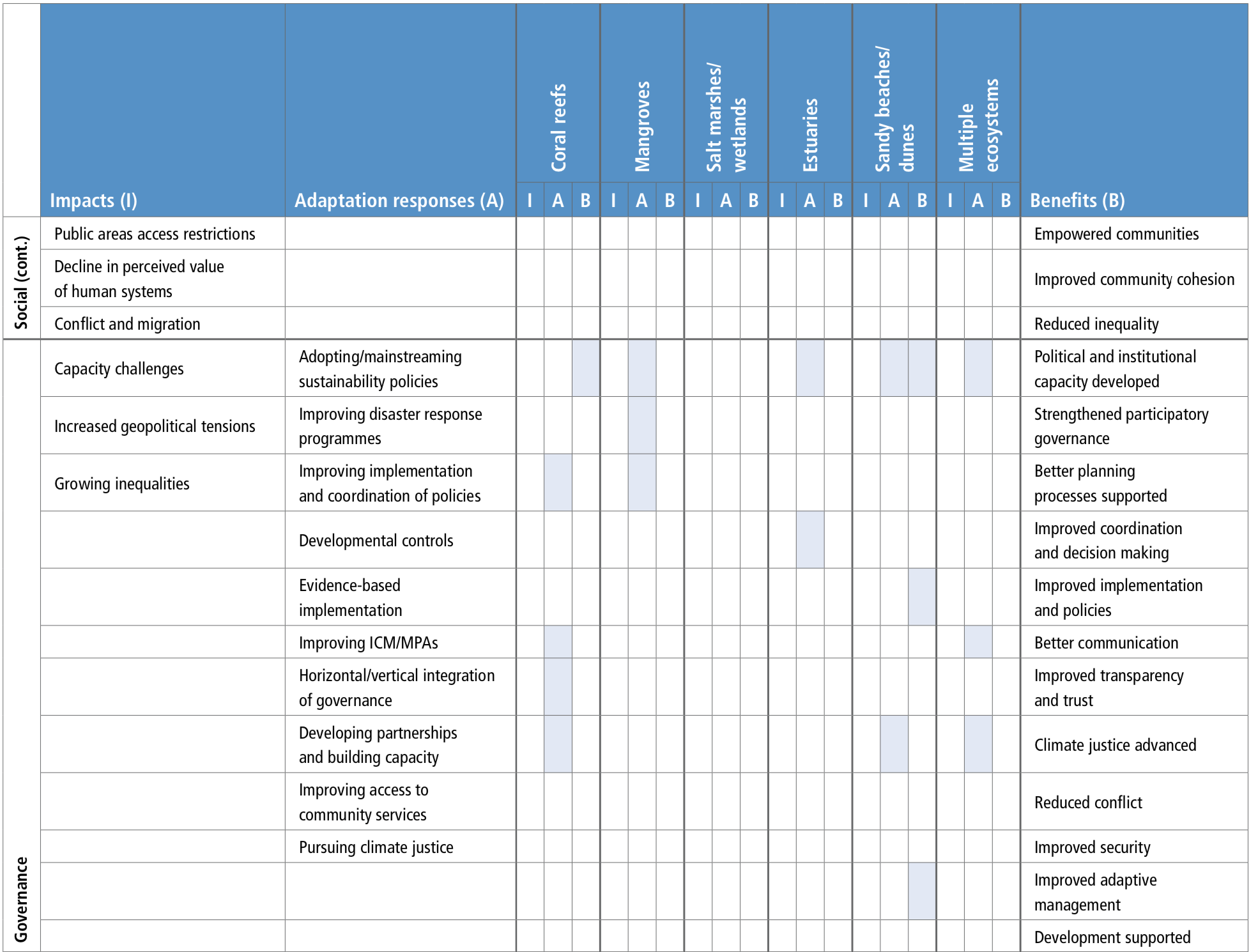
Socioinstitutional adaptation responses, including community-based adaptation, capacity-building, participatory processes, institutional support for adaptation planning and support mechanisms for communities are important tools to address climate change impacts (high confidence). For fisheries management, improving coordination of integrated coastal management and marine protected areas (MPAs) have emerged in the literature as important adaptation governance responses (robust evidence, medium agreement).{5.5.2.2, 5.5.2.6}
Observed widespread decline in warm water corals has led to the consideration of alternative restoration approaches to enhance climate resilience. Approaches, such as ‘coral reef gardening’ have been tested, and ecological engineering and other approaches such as assisted evolution, colonisation and chimerism are being researched for reef restoration. However, the effectiveness of these approaches to increase resilience to climate stressors and their large-scale implementation for reef restoration will be limited unless warming and ocean acidification are rapidly controlled (high confidence). {Box 5.5, 5.5.2}
Existing ocean governance structures are already facing multi-dimensional, scale-related challenges because of climate change. This trend of increasing complexity will continue (high confidence). The mechanisms for the governance of marine Areas Beyond National Jurisdiction (ABNJ), such as ocean acidification, would benefit from further development (high confidence). There is also scope to increase the overall effectiveness of international and national ocean governance regimes by increasing cooperation, integration and widening participation (medium confidence). Diverse adaptations of ocean related governance are being tried, and some are producing promising results. However, rigorous evaluation is needed of the effectiveness of these adaptations in achieving their goals. {5.5.3}
There are a broad range of identified barriers and limits for adaptation to climate change in ecosystems and human systems (high confidence). Limitations include the space that ecosystems require, non-climatic drivers and human impacts that need to be addressed as part of the adaptation response, the lowering of adaptive capacity of ecosystems because of climate change, and the slower ecosystem recovery rates relative to the recurrence of climate impacts, availability of technology, knowledge and financial support and existing governance structures (medium confidence). {5.5.2}